After two and a half years providing COVID-19 testing services to New England and beyond – including over 1.6 million tests – JAX’s Clinical Genomics Laboratory has suspended COVID-19 testing as of Friday, July 29, 2022, in alignment with the significant decrease in COVID-19 PCR testing demand. We will continue to monitor the pandemic and testing needs, and are prepared to reinstate testing services should demand increase. We are grateful to our community partners for supporting and collaborating with the JAX CLIA lab on this important endeavor.
While testing and sequencing services are no longer JAX’s focus at present, we are continuing our vital COVID-19 research, including the investigating the biological complexities of COVID, long COVID, and developing mouse models for the study of this virus.
The JAX CLIA lab’s focus has now returned to serving the oncology community with complex tumor profiling assays that help cancer patients and their physicians learn about specific tumors and provide insight into the most promising treatment options. We are proud of the continued role we play in improving human health and empowering the global biomedical community in our shared quest to improve human health.

Researchers at The Jackson Laboratory and Trudeau Institute have identified the first mouse strain that is susceptible to severe COVID-19 without the need for genetic modification.
View more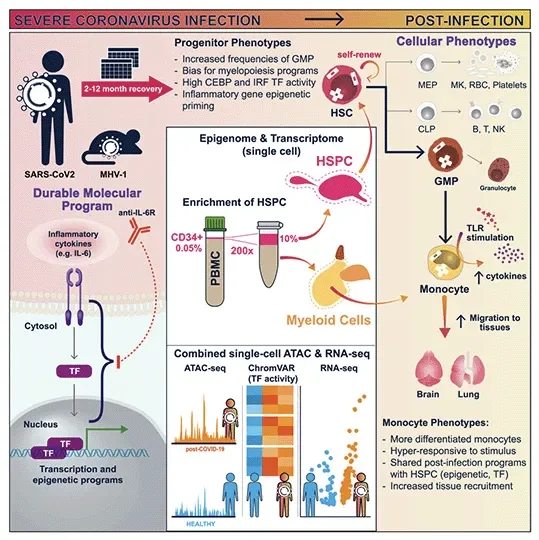
Severe COVID-19 triggers changes that affect gene expression in immune system stem cells, causing long-lasting alterations in the body’s immune response, according to a new study by Weill Cornell Medicine and Jackson Laboratory investigators. The finding could help explain symptoms of prolonged inflammation and long COVID in people who have had the disease.
View more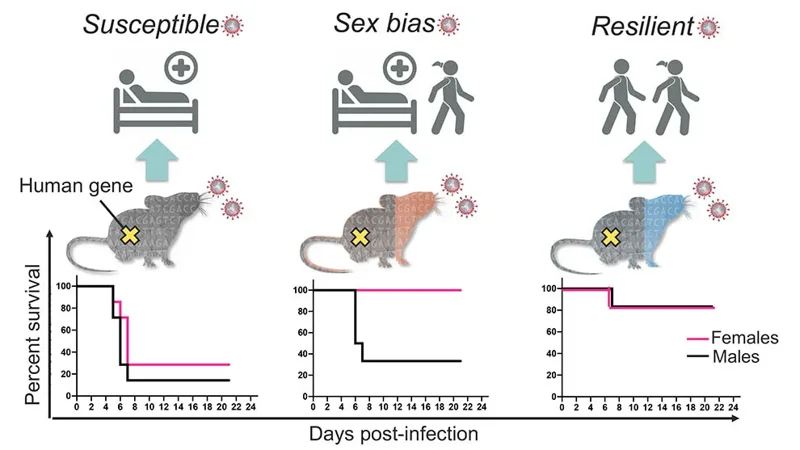
Researchers at The Jackson Laboratory have created a panel of genetically diverse mice that accurately model the highly variable human response to SARS-CoV-2 infection. Together with collaborators at NIH’s Rocky Mountain Laboratories, the team uncovered differences in the innate immune and regulated proinflammatory responses, the timing and strength of which are associated with disease severity. Moving forward, the diverse mouse strains will allow scientists to model patient variation in COVID-19 outcome and provide a platform for discovering biomarkers of disease severity, characterizing antiviral immune responses and evaluating countermeasures.
View more
Over the past three years, it has become clear that, for many people, the effects of SARS-CoV-2 infection persist long after an initial case of COVID-19 wanes.
View moreBuilding a mouse squad against Covid-19: It began with an email from Wuhan, a Maine laboratory and mouse sperm from Iowa. Now that lab is on the verge of supplying a much-needed animal for SARS-CoV-2 research. Read on Knowable
From Hamsters to Baboons: The Animals Helping Scientists Understand the Coronavirus: Different species are helping answer different questions about COVID-19 in humans in order to develop vaccines and treatments. Read on Scientific American
Mice, hamsters, ferrets, monkeys. Which lab animals can help defeat the new coronavirus?: Scientists hope that a global, cooperative rush to create lab animals that mimic COVID-19 when infected will lead to drugs or a vaccine that can stop the pandemic. Read on Science
Researchers are rushing to freeze… lab mice sperm?: With COVID-19 closing research labs, scientists are putting lab mice sperm on ice to save science. Read on Freethink

Your gift makes it possible for discoveries to happen faster, for novel ideas to be explored and for research to accelerate forward at a scale that delivers life-changing scientific breakthroughs.
View more