Antibody therapeutics offer incredible versatility for therapeutic design and are the basis of an array of approved treatments for diseases in cancer, autoimmunity, neurobiology, and metabolic disorders. The advantage of this approach is our ability to exploit the natural ability of cells to create hundreds of thousands of antibodies and select the ones with the strongest affinity for their target(s), to block or to activate them. However, the preclinical development of antibodies poses unique challenges as antibodies display a high level of species-specificity not only in target binding through the variable region but also in the Fc region, which is required for extending half-life. Your success depends on accurate analysis of pharmacokinetic (PK) factors that impact your drug candidate(s) performance.

Translationally Relevant
Humanized FcRn mice provide PK data comparable to nonhuman primate and human values using allometric scaling.

Simple, quick and cost-effective solution
Simple study design that can be executed within 3-4 week periods and cost significantly less than NHP studies.

Advancing Therapies from Bench to Bedside
The HuPK platform improves candidate selection for researchers, boosting success rates and speeding up therapeutic development.
The HuPK platform out performs other murine models such as wildtype mice and FcRn knock-out mice to produce more translationally-relevant PK data. Through hundreds of PK/PD studies by JAX Preclinical Services, and from dozens of publications, over the past 20+ years, Tg32 mice have demonstrated that human FCGRT gene expression in Tg32 mice yields data that translates exceptionally well to the clinic.
| Human predicted values from allometric scaling of Tg32 Hom Mice Data | |||
|---|---|---|---|
| Antibody | t1/2 | Clearance | Volume of Distribution at Steady State |
| Belatacept | 3.03 days | 0.327 ml/hour.kg | 41.24 ml/kg |
| Ipilimumab | 15.07 days | 0.425 ml/hour.kg | 230.9 ml/kg |
| Pembrolizumab | 23.15 days | 0.069 ml/hour.kg | 58.43 ml/kg |
Get an introduction to JAX's HuPK platform. Learn about the main applications such as:
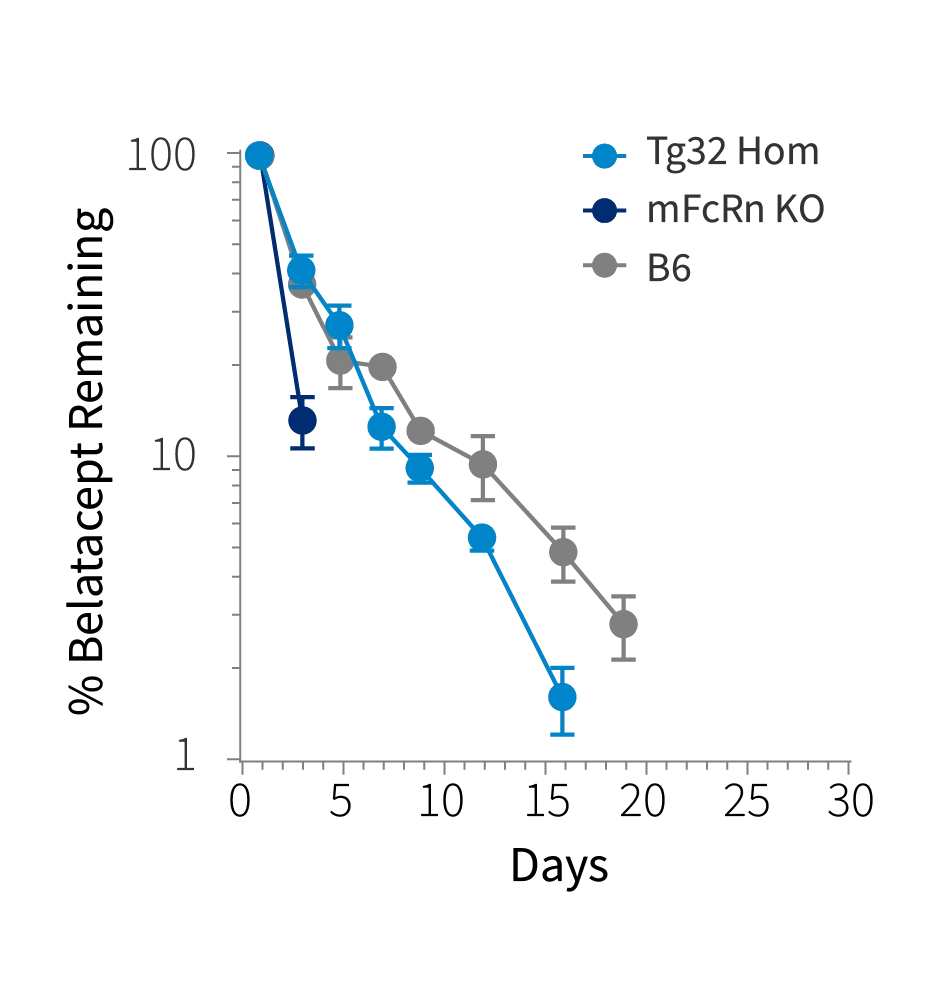
Figure 1a
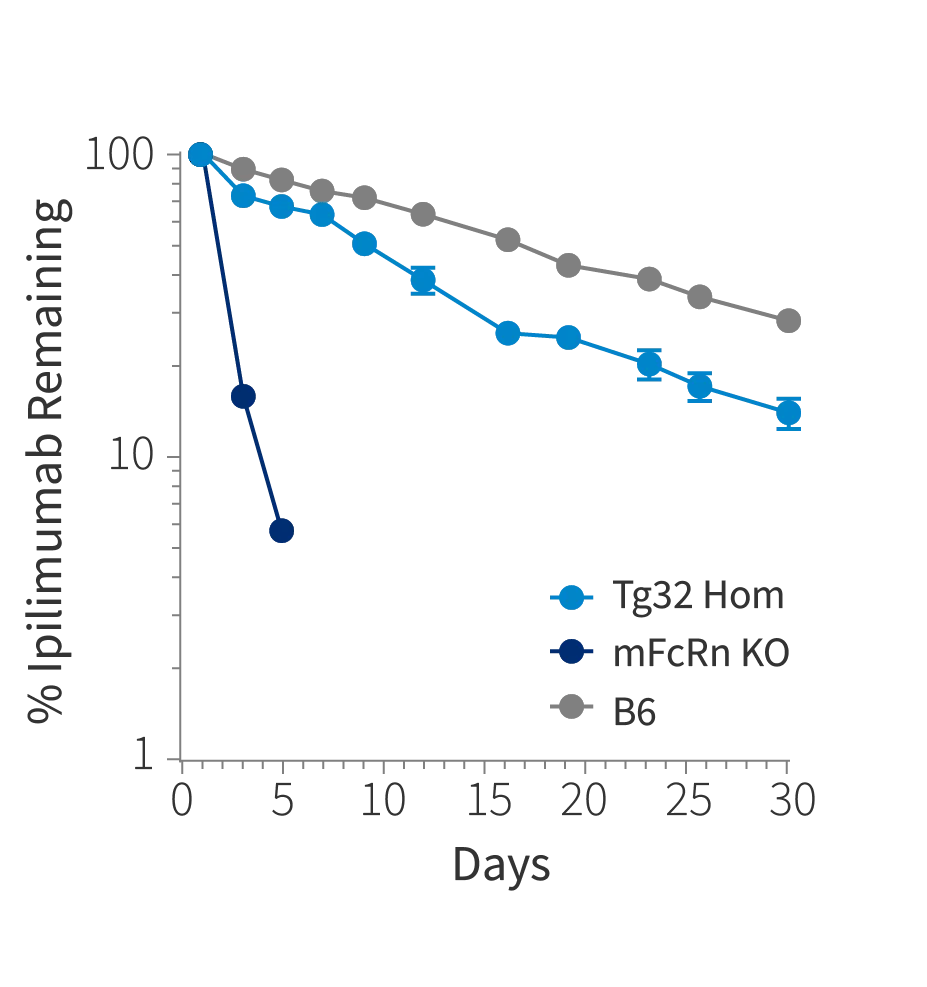
Figure 1b
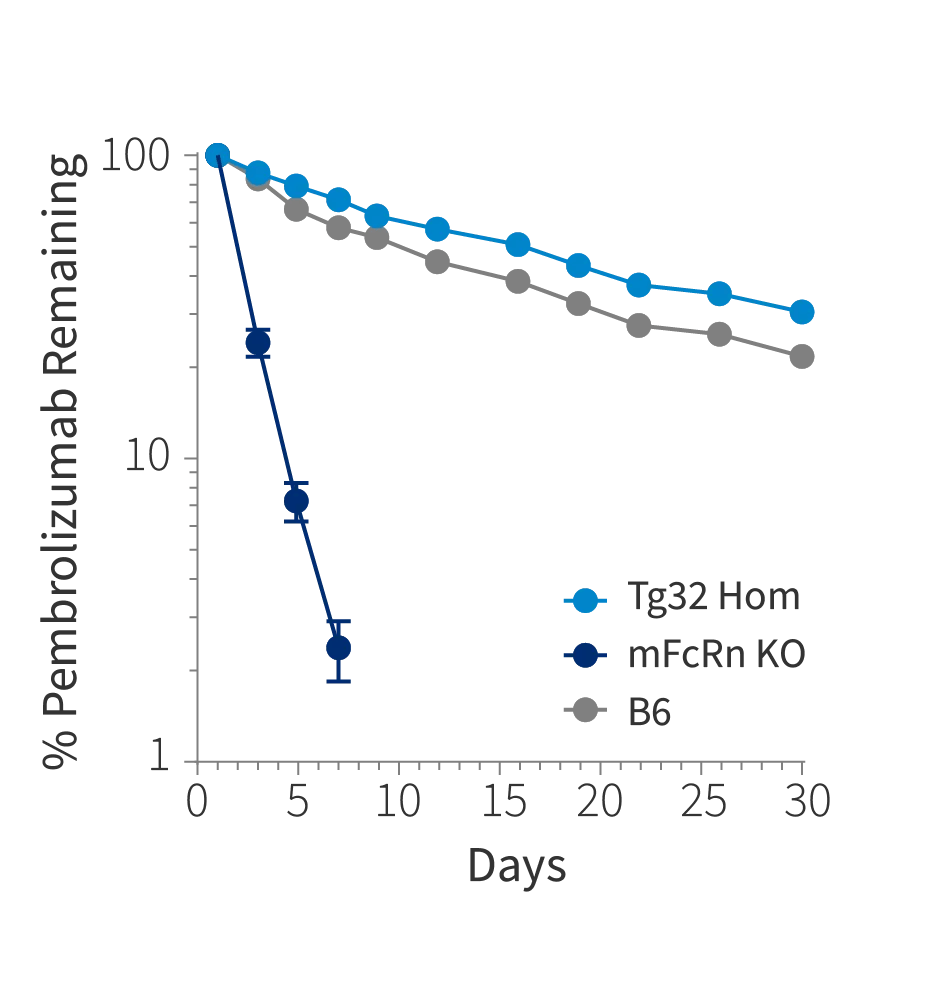
Figure 1c
Figure 1: Plasma concentration time plot of a) Belatacept, b) Ipilimumab and c) Pembrolizumab at single intravenous dose at 10mg/kg in Tg32 Hom (014565), mFcRn KO mice and C57BL/6J mice (000664)
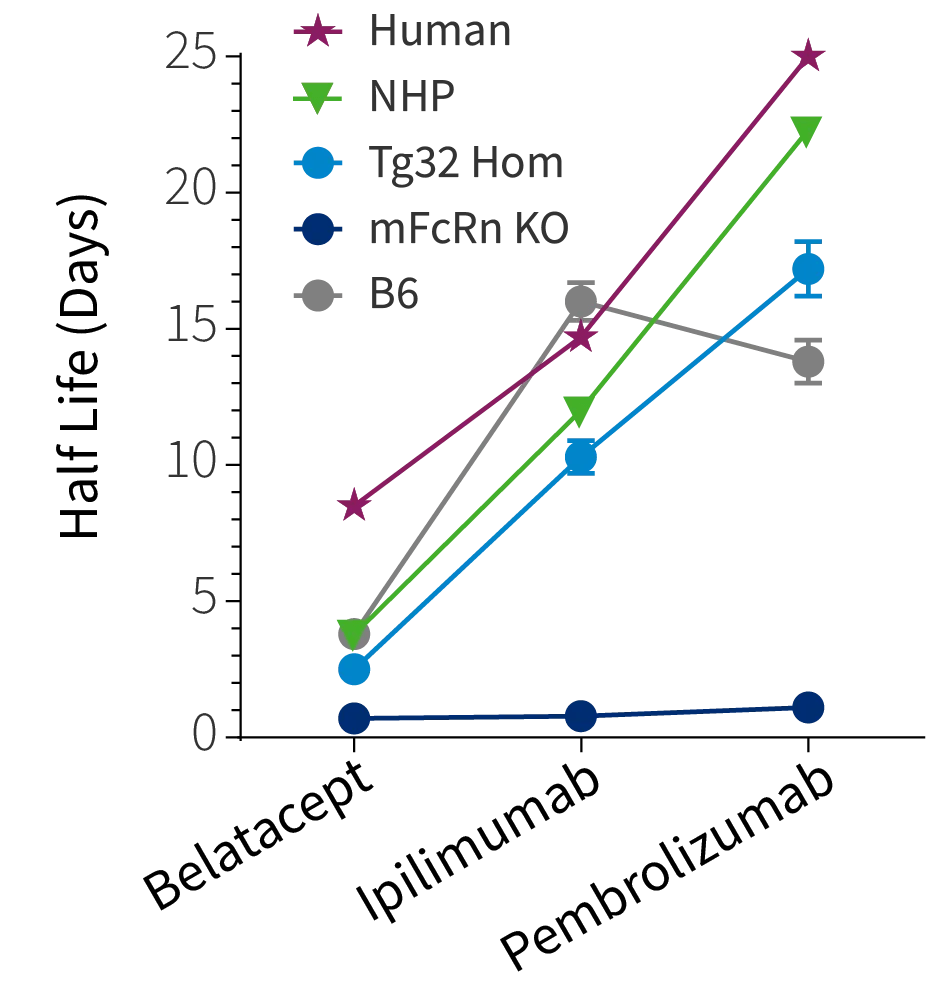
Figure 2a
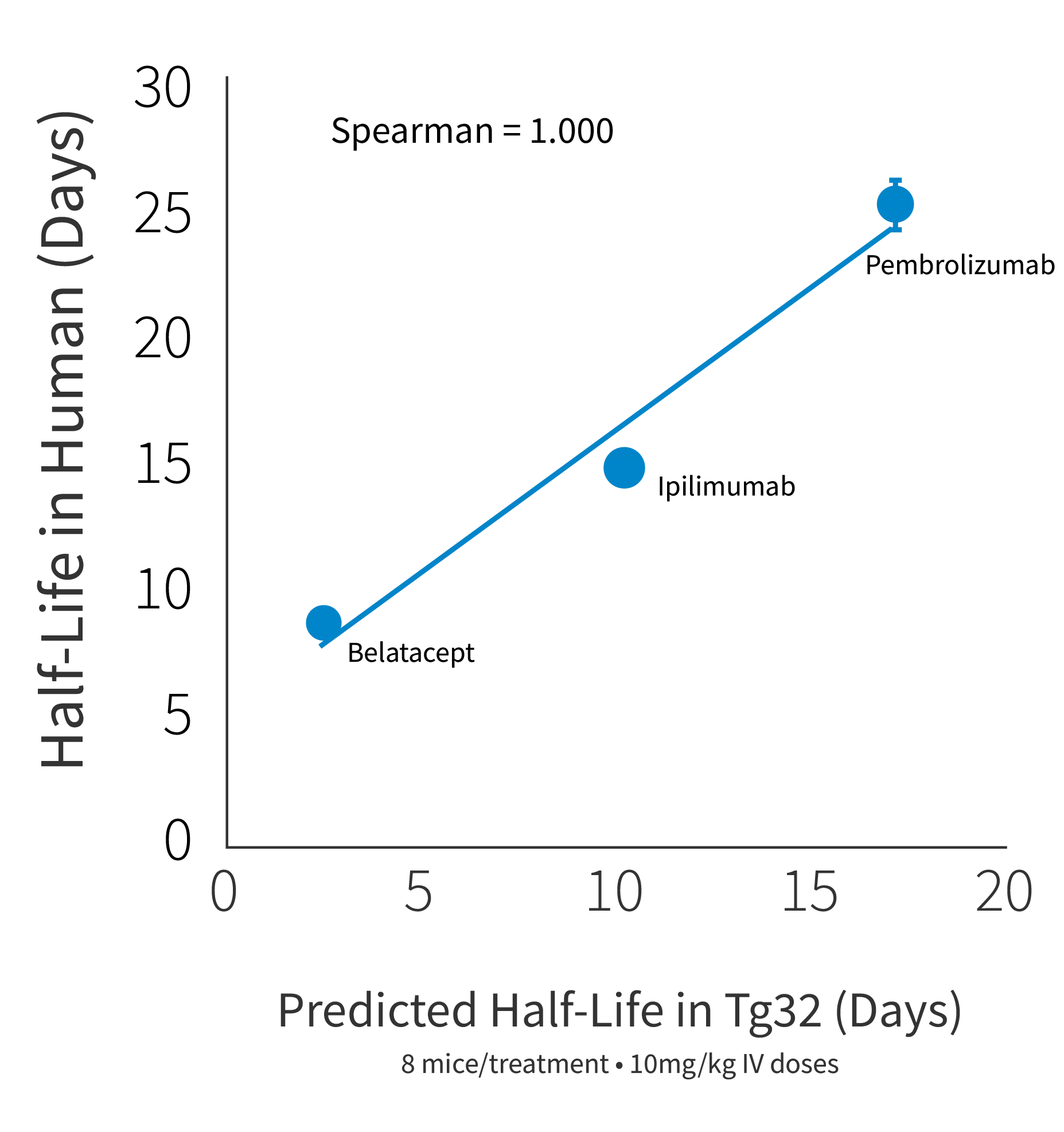
Figure 2b
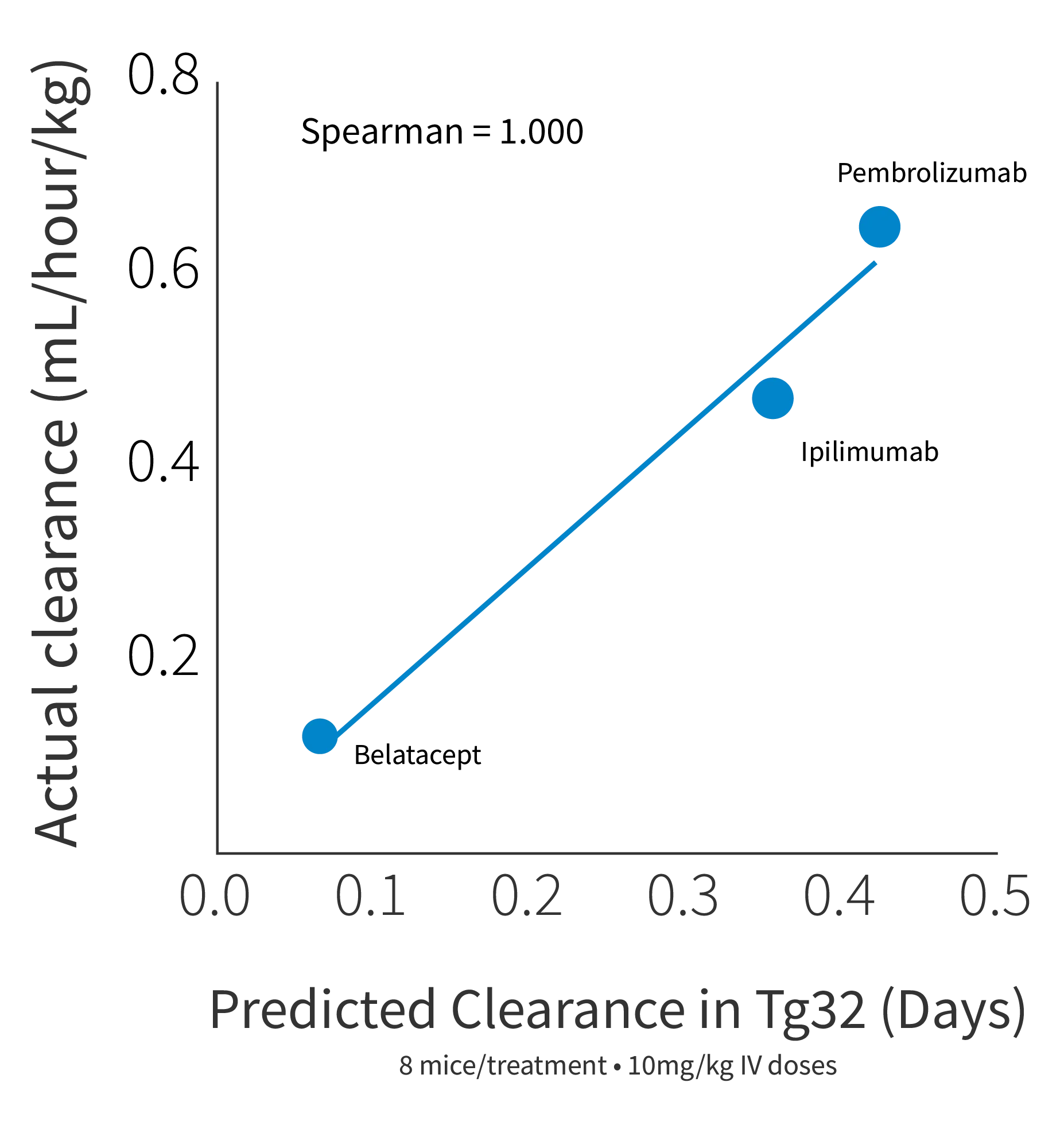
Figure 2c
Figure 2: a) Half life curve of Belatacept, Ipilimumab and Pembrolizumab in Tg32 Hom mice (014565) correlates well to the corresponding NHP and human half life's of these antibodies. b) Predicted human half life from Tg32 Hom mice through allometric scaling perfectly correlated with actual human half life for 3 antibodies. c) Predicted human clearance value from Tg32 Hom mice through allometric scaling perfectly correlated with actual human clearance data.
Let our experts help you select a model to address anti-drug antibody (ADA) response, human albumin conjugation, half life extended antibodies or more refined PK data to assist in lead selection.
| Model | Characteristics | Main Application | Model Features |
|---|---|---|---|
| hFcRn Tg32 (014565) | The most validated model on the market for therapeutic antibody PK prediction. | Prediction of the clinical half-life of therapeutic antibodies through allometric scaling. | Physiological expression of the human FCGRT gene coding for the FcRn protein. |
| hFcRn Tg32 SCID (018441) | No ADA response to humanized molecules, allowing PK and PD evaluation of highly engineered molecules. | Prediction of the clinical half-life of antibody-based molecules with strong immunogenicity in mice (i.e., Fc-fusion bispecific antibodies.) | Physiological expression of the human FCGRT gene and lack of mature B cells. |
| Tg32-hFc (029686) | Expression of human IgG1 in place of mouse IgG1. | Provides concentration steady state human IgG1 as a PD readout when FcRn is therapeutically targeted. | Physiological relevant expression of human IgG1. |
| hFcRn Tg276 (004919) | Lower FcRn function compared to Tg32 that is highly correlated with human PK outcomes. | Provides PK differentiation for test molecules not distinguished by Tg32, and shorter in-life PK studies especially for long-lived Fc variants. | Ubiquitous expression of the human FCGRT gene coding for the FcRn protein. |
| NSG hFcRn Tg32 (028615) | Human FcRn Tg32 expression in mice routinely engrafted with human cells. | These Tg32 mice can be engrafted with human CD34+ hematopoietic stem cells (HSC), peripheral blood mononuclear cells (PBMC), patient derived xenografts (PDX), or adult stem cells and tissues. | While PK yielded correlates with human PK, FcRn function is diminished due to unique FcgR expressed by NSG. |
| hFcRn Tg32 Alb KO (025201) | Features mouse albumin deficiency and human FcRn Tg32 function. | Characterizes PK of human albumin-formatted therapeutics in the absence of high affinity mouse albumin. | KO of mouse Alb gene removes this high affinity competition for human FcRn protection of albumin-formatted therapeutics. |
| hFcRn Tg32 hAlb (037524) | Model expresses both human FcRn and human serum albumin. | Evaluate PK of albumin-formatted drugs in the presence of endogenous human albumin, and off-target PD responses of FcRn targeted therapies. | Model express human serum albumin at physiological steady state 25 to 40 mg/ml. |
Learn about the integration of humanized FcRn mice in Roche's drug development pipeline to assess safety and efficacy of a bispecific antibody in a Phase 1 clinical trial.
Not what you're looking for?
Visit our Resource Hub