The NSG-SGM3 mouse is a revolutionary in vivo model that allows exceptional engraftment of human tissues including hematopoietic cells. Learn which human cytokines support remarkable engraftment of diverse hematopoietic lineages, primary human AML samples and multiple cancer types compared to other existing models.
What makes NSG-SGM3 a revolutionary strain?
Blog Post | March 2, 2016
The NSG™-SGM3 transgenic strain is a remarkable in vivo model generated by Dr. James Mulloy (Cincinnati Children's Hospital Medical Center). It was designed to improve the growth and engraftment of human leukemias – particularly those that have been very challenging to grow in other immunodeficient strains. Since 2010, NSG-SGM3 mice have been used to better understand how human leukemias get started and subsequently develop and for testing novel therapeutic compounds for treating this devastating disease.
More recently, NSG-SGM3 mice have been used in the growing field of immuno-oncology to understand the interactions between “normal” human immune cells and specific patient-derived tumors. They are a robustin vivo platform for analyzing the safety and effectiveness of potential new drugs to hematologic diseases, human infectious diseases, and most types of human cancers.
What is so special about the NSG-SGM3 mouse model?
1. NSG genetic background
NSG-SGM3 mice were developed on the NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) background, and like their NSG parents, NSG-SGM3 mice lack mature T cells, B cells, and functional NK cells, and are deficient in mouse cytokine signaling. These characteristics are required to support the ready and stable engraftment of human CD34+ hematopoietic stem cells, and have made NSG mice a superior, long-lived host for studies employing xenotransplantation strategies.
2. Human hematopoietic cytokines
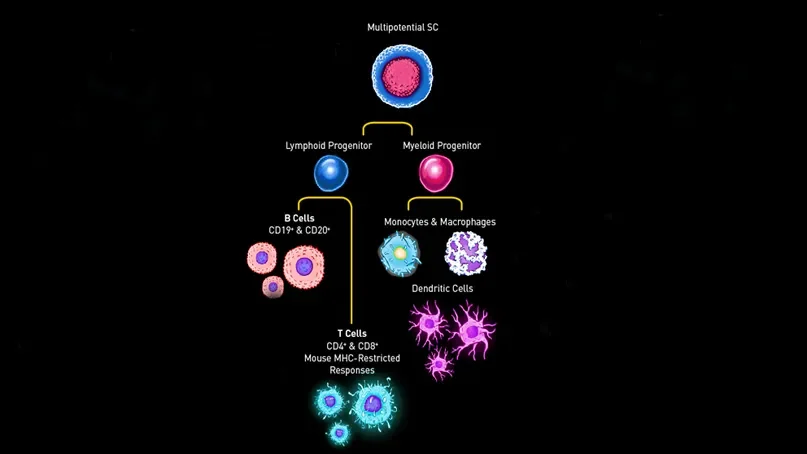
Cytokines play a major role in stimulating immune cell hematopoiesis. They are potent at extremely low concentrations and communicate with target cells by binding specific receptors. They mainly act upon immature cells by promoting their maturation and proliferation of daughter cells within particular hematopoietic lineages.
NSG-SGM3 mice contain three co-injected transgenes, each driven by a human cytomegalovirus promoter/enhancer sequence. Each transgene is represented in the strain’s “SGM3” nickname:
S: Human Stem Cell Factor (SCF) gene, also known as KIT ligand (KITLG), acts on hematopoietic stem cells to accelerate their entry into the cell cycle, and is required to maintain normal basal hematopoiesis.
GM: Human Granulocyte/Macrophage-colony stimulating factor 2 (GM-CSF), also known as CSF2, increases neutrophil, eosinophil, and monocyte/macrophage numbers and function.
3: Human interleukin-3 (IL-3) stimulates multipotent hematopoietic stem cells to differentiate into myeloid progenitor cells. In addition, it stimulates all cells in the myeloid lineage (granulocytes, monocytes, and dendritic cells) to proliferate in conjunction with other cytokines.
Triple transgenic NSG-SGM3 mice constitutively produce 2-4 ng/ml serum levels of human SCF, GM-CSF, and IL-3, providing cell proliferation and survival signals that become more measurable over time. These mice support the stable engraftment of CD33+ myeloid lineages including several types lymphoid cells. As a result, NSG-SGM3 mice allow superior engraftment of diverse hematopoietic lineages and, importantly, primary human AML samples compared to other existing models.
Compared to the humanized NSG™ model, humanized NSG™-SGM3 mice have increased cell counts of the following human immune cell populations:
- CD33+ myeloid cells
- Hematopoietic stem cells
- Myeloid progenitor cells
- Mast cells
- Myeloid dendritic cells
- B cells (CD19+)
- T cells (CD3+)
- T helper cells (CD4+)
- T cytotoxic cells (CD8+)
- Treg cells (CD4+, CD25+, FoxP3+)
3. Improved AML xenograft efficiency
Despite major advances in human mouse xenografts, most patient-derived acute myeloid leukemia (AML) samples fail to adequately engraft in most of the commonly used immunodeficient mouse strains. The reasons for this include poor homing to the bone marrow, failure to survive in the foreign bone marrow niche, and limited expansion in the absence of specific human growth factors. Primary AML samples that fail to engraft in other mouse models successfully engraft in NSG-SGM3 mice, even when the cells available from a human AML donor are limited (Wunderlich et al 2010).
JAX currently has experiment-ready AML engrafted mice and cohorts of NSG-SGM3 mice readily available for your studies.Ordering cohorts of these mice with high health status and expanded availability is a lot more convenient than maintaining this highly immunodeficient strain at your facility.
4. In vivo platforms for Immuno-oncology
NSG-SGM3 mice can be co-engrafted with both human hematopoietic stem cells that differentiate into human myeloid cells and lymphoid cells and with patient-derived xenografts. This humanized NSG-SGM3-PDXin vivo model more closely simulates the conditions in human cancer patients with functioning immune systems, and is an outstanding tool for therapeutic testing of novel immunotherapies that modulate the human immune system to help destroy cancers.
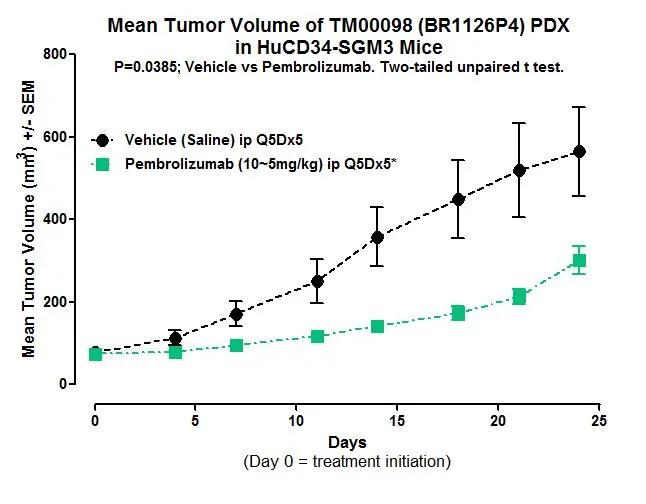
Human CD34+ hematopoietic stem cell-engrafted NSG-SGM3 mice (hu-CD34) are a validated platform for immuno-oncology efficacy studies, and infectious disease research. JAX In Vivo Pharmacology Services recently presented exciting data demonstrating tumor growth restriction and inhibition in hu-NSG-SGM3-PDX mice following treatment with immune checkpoint inhibitors. “The NSG-SGM3 model is unique in that it offers the most physiological co-engraftment of patient tumors and human immune cells, including myeloid cells that are critically important in modulating human responses to cancers,” stated James Keck, Ph.D., JAX Senior Director of In Vivo Pharmacology Services.
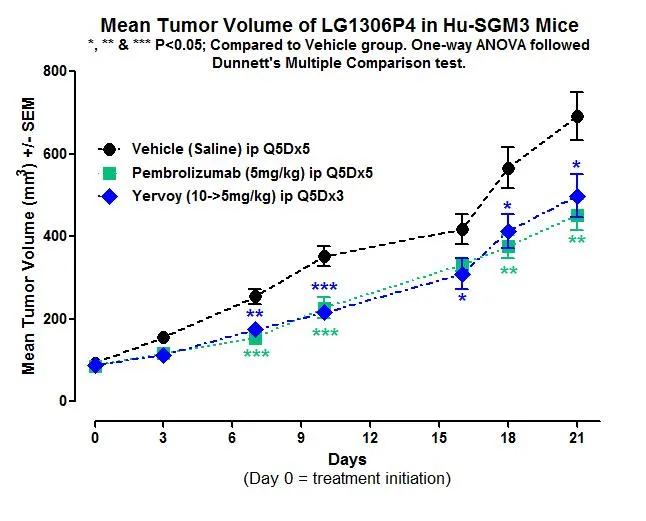
Results in Figure 1 highlight the potential for use of this cutting-edge platform for pre-clinical efficacy testing of immuno-oncology compounds.
The NSG™-SGM3 mouse represents an evolution of the NSG model that allows exceptional engraftment of human tissues including hematopoietic cells, AML primary samples, and multiple cancer types. Physiologically, hu-NSG-SGM3-PDX mice more closely simulates the biological mileu within human cancer patients, and is being actively used to develop cancer immunotherapies. Our large inventory has CD34+ mice available for immediate shipment to your facility or efficacy study enrollment at ours.
When considering the best model(s) for your oncology or immuno-oncology study, consider working with JAX® Preclinical Services Team to access our decades of experience executing complex preclinical studies that deliver translational proficiency - advancing your research faster and with less risk. Talk with us about your project, your timelines, and options that align with your budget.