
From 2002 to 2012, the NIH funded the Reproductive Genomics Program at the Jackson Laboratory for Mammalian Genetics (Bar Harbor), run by Drs. John J. Eppig, Mary Ann Handel, and John Schimenti. The overall goals of this program were to produce and distribute mutant models for infertility and to investigate underlying causes in order to identify new genes and pathways contributing to reproductive function.
ENU-mutagenesis of male mice was followed by a standard three-generation breeding scheme, with fertility testing of all G3 offspring in order to identify putative reproductive mutants.
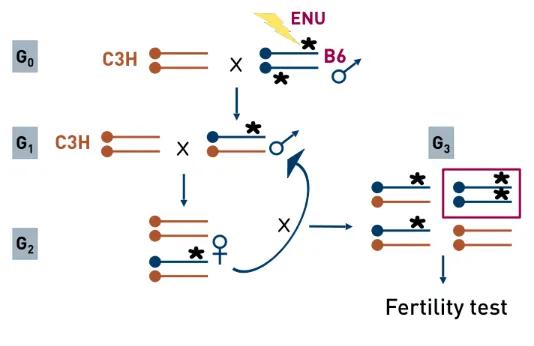
In all, almost 500 pedigrees were established; approximately 20,000 individual G3 mice were screened by phenotype analyses, and close to 50 new mutations were identified. Phenotype screening protocols covered basic aspects of reproductive histology and physiology.
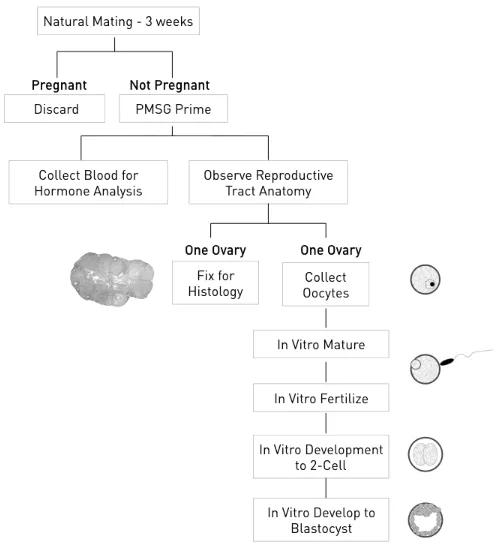
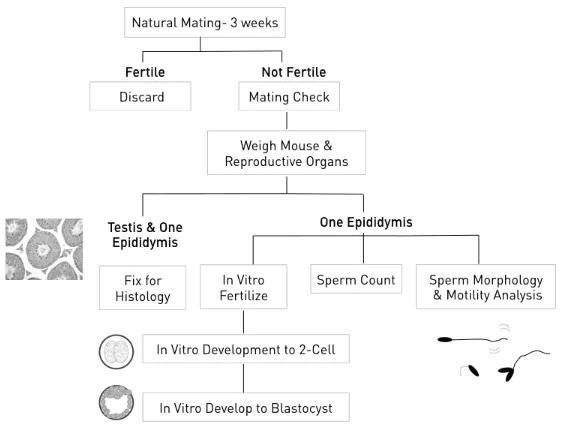
Of the mutant lines established, about 75% exhibited a male infertility phenotype only, almost 10% exhibited female infertility only, and about 15% exhibited infertility of both sexes. All mutations were mapped to a chromosome region and all were archived as cryopreserved sperm from heterozygous males. Of these, the following mutations listed in Table 1 were fine mapped and the affected genes identified.
Mutant Line | Affected Gene |
|---|---|
repro1 | Iqcg |
repro4 | Mtap2 |
repro5 | Brwd1 |
repro7 | Prdm9 |
repro8 | Eif4g3 |
repro9 | Mybl1 |
repro11 | Smc1b |
repro12 | Akap9 |
repro22 | Mad2l2 (Rev7) |
repro23 | Tdrd12 |
repro27 | Golga3 |
repro32 | Capza3 |
repro34 | Stx2 |
repro42 | Spata22 |
repro57 | Rnf212 |
ferf1 | Dnahc1 |
Marf1 | 4921513D23Rik |
omd1 | Padi6 |
spcar3 | Setx |
vac* | Chp1 |
Ward, J, L Reinholdt, S Hartford, L Wilson, R Munroe, K Schimenti, B Libby, M O’Brien, J Pendola, JJ Eppig, J Schimenti. 2003. Towards the genetics of mammalian reproduction: induction and mapping of gametogenesis mutants in mice. Biol Reprod 69:1615-25.
Lessard C, JK Pendola, SA Hartford, JC Schimenti, MA Handel, JJ Eppig. 2004. New mouse genetic models for human contraceptive development. Cytogenet Genome Res 105:222-227.
Handel, MA, C Lessard, L Reinholdt, J Schimenti, JJ Eppig. 2006. Mutagenesis as an unbiased approach to identify novel contraceptive targets. Mol Cell Endo 250:201-205.
Bannister L, R Pezza, J Donaldson, D de Rooij, K Schimenti, D Camerini-Otero, JC Schimenti. 2007. Male-specific sterility in mice carrying a dominant, recombination-defective allele of the RecA homologDmc1. PLoS Biol 5:e105. doi:10.1371/journal.pbio.0050105. PMID: 17425408.
Furnes B, JC Schimenti. 2007. Fast forward to new genes in mammalian reproduction. J Physiol 578:25-32. doi: 10.1113/jphysiol.2006.119164.PMID: 16973708.
Lessard C, H Lothrop, JC Schimenti, MA Handel. 2007. Mutagenesis-generated mouse models of human infertility with abnormal sperm. Hum Reprod 22:159-166.
Akiyama K, S Akiyama, Y Asano, M Khalaj, C Kiyosu, AA Masoudi, S Takahashi, K Katayama, T Tsuji, J Noguchi, T Kunieda. 2008. A new ENU-induced mutant mouse with defective spermatogenesis caused by a nonsense mutation of the Syntaxin 2/Epimorphin (Stx2/Epim) gene. J Reprod Dev 54:122-128.
Khalaj M, AE Abassi, R Nishimuta, K Akiyama, T Tsuji, J Noguchi, T Kunieda. 2008. Leydig cell hyperplasia is an ENU-induced mutant mouse with germ-cell depletion J Reprod Dev 54:225-228.
Philipps, D., K Wigglesworth, S Hartford, F Sun, S Pattabiraman, K Schimenti, MA Handel, JJ Eppig, J Schimenti. 2008. The dual bromodomain and WD repeat-containing mouse protein BRWD1 is required for normal spermiogenesis and the oocyte-embryo transition. Devel. Biol. 317:72-82.
Asano Y, K Akiyama, T Tsuji, S takahashi, J Noguchi, T Kunieda. 2009. Characteriztion and linkage mapping of an ENU-induced mutant mouse with defective spermatogenesis. Exp. Anim. 58:525-532.
Geyer CB, AL Inselman, JA Sunman, S Bornstein, MA Handel, EM Eddy. 2009. A missense mutation in theCapza3 gene and disruption of F-actin organization in spermatids of repro32 infertile male mice. Dev Biol 330:142-152.
Handel MA, JC Schimenti. 2010. Genetics of mammalian meiosis: regulation, dynamics and impact on fertility. Nat Rev Genet 11:124-136.
Li S, A Francisco, C Han, S Pattabiraman, M Foote, S Giesy, C Wang, J Schimenti, Y Boisclair, Q Long. 2010. The full-length Isoform of pleckstrin homology domain-interacting protein (PHIP) is required for postnatal growth.FEBS Letters, 584:4121-4127. PMCID: PMC2965186
Sun F, K Palmer, MA Handel. 2010. Mutation of Eif4g3, encoding a eukaryotic translation initiation factor, causes male infertility and meiotic arrest of mouse spermatocytes. Development 137:1699-1707.
Bolcun-Filas, E., L Bannister, A Barash, K, S Hartford, JJ Eppig, MA Handel, L Shen, J Schimenti. 2011. A-MYB is a master regulator of meiosis in male mice.Development 138:3319-3330.
Sun F, MA Handel. 2011. A mutation in Mtap2 is associated with arrest of mammalian spermatocytes before the first meiotic division. Genes 2(1):21-25. doi:10.3390/genes2010021
Bolcun-Filas E, J Schimenti. 2012. Genetics of meiosis and recombination in mice. Intl Rev Cell Molec Biol 298:179-227.
Guan Y, D Gorenshteyn, JC Schimenti, MA Handel, CJ Bult, MA Hibbs, OG Troyanskaya. 2012. Tissue-specific functional networks for prioritizing disease genes. PloS Comp Biol 8:e1002694.
La Salle S., K Palmer, M O'Brien, J Schimenti, JJ Eppig, MA Handel. 2012.Spata22, a novel vertebrate-specific gene, is required for meiotic progress in mouse germ cells. Biol Reprod 86:45. PMID22011390
Su, Y-Q, K Sugiura, F Sun, J Pendola, G Cox, MA Handel, J Schimenti, JJ Eppig. 2012. MARF1 regulates essential oogenic processes in mice.Science, 335:1496-9.
Su, Y-Q, F Sun, MA Handel, JC Schimenti, JJ Eppig. 2012. Meiosis arrest female 1 (MARF1) has nuage-like function in mammalian oocytes. Proc Natl Acad Sci USA 109:18653-18660.
Bentson, LF, VA Agbor, LN Agbor, AC Lopez, LE Nfonsam, SS Bornstein, MA Handel, CC Linder. 2013. New point mutation inGolga3 causes multiple defects in spermatogenesis. Androl 1:440-450.
Fujiwara, Y, N Ogunuke, K Inouye, A Ogura, MA Handel, J Noguchi, T Kunieda. 2013. t-SNARE Syntaxin2 (STX2) is implicated in intracellular transport of sulfoglycolipids during meiotic prophase in spermatogenesis. Biol Reprod 88:141, 1-9.
Li, X, C Roy, X Dong, E Bolcun-Filas, J Wang, B Han, J Xu, M Moore, J Schimenti, Z Weng, P Zamore. 2013. An ancient transcription factor initiates the burst of piRNA production during meiosis in the mouse testis. Molec Cell 50:67-81.
Liu Y, HC Zaun, J Orlowski, SL Ackerman. 2013. CHP1-mediated NHE1 biosynthetic maturation is required for Purkinje cell axon homeostasis. J Neurosci 33:12656-12669.
Luo M, F Yang, NA Leu, J Landaiche, MA Handel, R Benavente, S La Salle, PJ Wang. 2013. MEIOB exhibits single-stranded DNA-binding and exonuclease activities and is essential for meiotic recombination. Nat Commun 2013 Nov 18; 4:2788.
Pandey RR, Y Tokuzawa, E Hayashi T Ichisaka, S Kajita, Y Asano, T Kunieda, R Sachidanandam, S Chuma, S Yamanaka, RS Pillai. 2013. Tudor domain containing 12 (TRD12) is essential for secondary PIWI interacting RNA biogenesis in mice. Proc Natl Acad Sci 110:16492-16497.
Schimenti K, S Feuer, L Griffin, N Graham, C Bovet, S Hartford, J Pendola, C Lessard, J Schimenti, J Ward. 2013. AKAP9 is vital for spermatogenesis and Sertoli cell maturation in mice. Genetics194:447-457.
Harris T, K Schimenti, R Munroe, J Schimenti. 2014. IQ motif-containing G (Iqcg) is required for mouse spermiogenesis. G3: Genes, Genome, Genetics4:367-372. doi:10.1534/g3.113.009563. PMID: 24362311.
Abbasi A, M Khalaj, K Akiyama, Y Mukai, H Matsumoto, N Said, M Yoshida, T Kunieda. 2015. Lack ofRev7 function results in development of tubulostromal adenomas in mouse ovary. Mol Cell Endocrinol 412:19-25.
Fujiwara Y, H Matsumoto, K Akiyma, A Srivastava, M Chikushi, MA Handel, T Kunieda. 2015. An ENU-induced mutation in the mouseRnf212 gene is associated with male meiotic failure and infertility. Reproduction 149:67-74.
Khalaj M, A Abbasi, H Yamanishi, K Akiyama, S Wakitani, S Kikuchi, M Hirose, M Yuzuriha, M Magari, HA Degheidy, K Abe, A Ogura, H Hashimoto, T Kunieda. 2015. A missense mutation inRev7 disrupts formation of Polz impairing mouse development and repair of genotoxic agent-induced DNA lesions. J Biol Chem 289:3811-3824.
Pattabiraman S, C Baumann, D Guisado, JJ Eppig, JC Schimenti, R De La Fuente. Mouse BRWD1 is critical for spermatid postmeiotic transcription and female meiotic chromosome stability. 2015. J Cell Biol. 208(1):53-69.
Sun F, Fujiwara Y, Reinholdt LG, Hu J, Saxl RL, Baker CL, Petkov PM, Paigen K, MA Handel. 2015. Nuclear localization of PRDM9 and its role in meiotic chromatin modifications and homologous synapsis. Chromosoma Apr 18;PubMed PMID:25894966.