Diet restriction (DR) and HSC aging
Diet restriction (DR) is the "gold standard" of anti-aging interventions. Although the mechanisms of DR are unknown, we do know that it 1) increases maximum life span in a wide range of laboratory animals, including long-lived genotypes, while 2) delaying or preventing senescence for a very wide range of markers of aging, and 3) delaying the expression of almost all age-related pathology, including cancer. We were interested in the effects that DR would have specifically on HSC aging. Age-related loss of HSC function may be a major cause of anemias and can also lead to other defects. Treatments to improve this loss of function exist, but unfortunately, such treatments may increase the incidence of cancer.
We discovered that lifelong DR can mitigate the loss of HSC function with age; interestingly, this effect is strain dependent. Our discovery opens the door to the possibility that one of the ways DR retards aging is by counteracting genetically-regulated loss of stem cell function with age. Identifying the mechanism behind this effect could lead to treatment of aging stem cells that improves function while avoiding any increased risk of cancer.
DR alters aging in BALB/cByJ (BALB) hematopoietic stem cells (HSCs)
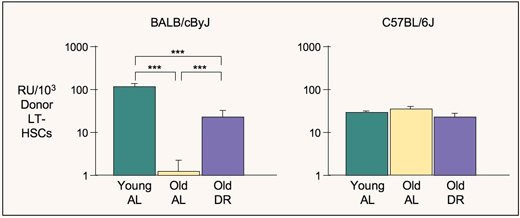
Starting at one month of age, we diet restricted BALB/cByJ (BALB) mice to 75% of the ad lib food intake of age- and sex-matched control BALB mice. Bone marrow cells (BMCs) from 25-month-old, diet restricted donors repopulated recipients nearly 3 times as well as BMCs from old controls, and about 1.7 times as well as BMCs from young controls (Chen et al., 2003).
Importantly, DR appears to prevent HSC proliferative exhaustion with aging by protecting clonal stability in HSCs from old BALB mice (Chen et al., 2003). The proportion of erythrocytes and lymphocytes in each recipient that was generated by HSCs from old diet restricted donors correlated well between 5 and 8 months after transplantation, with r = 0.95, so values at 8 months account for 90% of the variance at 5 months. This demonstrates clonal stability, because to sustain a constant proportion of donor-differentiated to competitor-differentiated cells in a given recipient, the same HSCs must be active. Clonal succession, the alternate, would produce no correlation over time.
Marrow from young BALB controls also showed clonal stability, with r = 0.97, accounting for 94% of the variance. Clonal stability was far less in marrow from old BALB controls, with r = 0.48, accounting for only 23% of the variance. This is consistent with HSC exhaustion.
To test if benefits of DR are due to a short-term response to low food intake independent of aging, we tested short-term DR in young BALB mice. DR for 5 months had far less effect than DR for 24 months. Short-term DR appeared to slightly improve HSC function, but differences were not significant. Thus, DR alters HSC aging, not a physiological set point, in the marrow stem cell compartment of BALB mice (Chen et al., 2003).
DR alters HSC aging differently in BALB/cByJ (BALB), C57BL/6J (B6), and BALBxB6 F1 hybrid mice
We studied the effects of DR on the ability of bone marrow cells to repopulate irradiated recipients and produce erythrocytes and lymphocytes in BALB, B6 and BALBxB6 F1 mice (Ertl et al., 2008). All control mice were matched by age, gender, and genotype. For a 3-month period starting at weaning, BALB mice were gradually diet restricted from 90% to 70% of the amount of food consumed by controls fed ad lib. From weaning, B6 and BALBxB6 F1 mice were diet restricted to 70% of the amount consumed by controls fed ad lib. Short-term, diet restricted mice were maintained on the 70% regimen until 6–7 months of age; long-term diet restricted mice remained on the 70% regimen until 22–25 months of age.
Results varied by strain. In BALB mice, repopulating abilities decline with age; DR ameliorates this trend. In B6 and BALBxB6 F1 mice, repopulating abilities increase with age; DR has no effect on this increase (Ertl et al., 2008; Ertl & Harrison, 2010).
Using antigenic markers to identify HSCs, we found that in aged BALB mice, HSC numbers are highly variable, and that the observed loss of marrow function results from a major loss in repopulating ability per HSC. DR ameliorates this loss. In B6 mice, repopulating ability per HSC is not affected by age or DR. Thus, DR increases or maintains increased marrow repopulating ability with age in the 3 genotypes tested, but effects on function depend on genotype. The findings that B6 repopulating ability per HSC does not change with age come from studies using whole marrow, in which repopulating abilities and numbers of HSCs are compared. When HSCs are enriched, those from old B6 mice do not function as well as those from young B6 mice, suggesting either damage due to enrichment, or change in markers with age, or both.
What the "little" dwarfing mutation in mice can teach us about the mechanisms of DR related to stem cell aging
Because DR reduces levels of insulin-like growth factor (IGF), reduces thyroid hormone (TH) responses, and increases corticosterone, we are testing whether one of these changes alone affects stem cell aging. We chose to study DR in the hyposomatotrophic "little" mouse (C57BL/6J-Ghrhrlit/J), homozygous for a mutation that removes function of the growth hormone releasing hormone receptor. The little mouse has reduced IGF but normal TH. HSC concentration per million cells appears to be doubled in young little mice compared to normal littermates (Sharma et al., 2005). We observe no deleterious effects on aging; in fact, the little mice outlive normal littermates by about 20%