Ovarian Transplantation to Overcome Breeding Difficulties
January 21, 2015

Some strains, despite all our efforts to minimize stress, optimize diets, and provide TLC, just don’t breed well. Their poor breeding performance may be due to particular mutations that limit their breeding productivity or affect their temperament or nurturing instincts. Moreover, the severity of such phenomena can be influenced by the strain’s genetic background. Fortunately, techniques are available to overcome such problems and improve the breeding performance of these troublesome colonies.
Ovarian transplantation circumvents poor breeding performance
Ovarian transplantation (OT) is a convenient method for overcoming breeding roadblocks introduced by mutations and transgenes. Strain B6CBA-Tg(HDexon1)62Gpb/1J (002810), for example, is one of the most commonly used mouse models for Huntington’s Disease with hemizygous (Tg/0) carriers of both sexes develop tremors and seizures by 9 to 11 weeks of age. Unfortunately, nearly all Tg/0 females and approximately 50% of Tg/0 males fail to produce even one litter. Further, of the males that do mate successfully, their breeding life span is limited to only a 3-4 week window. Maintaining a breeding colony of this strain using conventional matings, therefore, requires a very large number of breeding pairs that must be replaced frequently. The failure of Tg(HDexon1)62Gpb hemizygous females to breed is likely a consequence of their neurodegenerative phenotype and not due to a problem with their gametes. Indeed, oocytes produced in the hemizygous females’ ovaries are perfectly viable. Using ovarian transplantation, we can harvest the ovaries from hemizygote females, and engraft them into a histocompatible recipient female whose endogenous ovaries have been removed. Because the ovariectomized hosts do not carry the HDexon1 transgene in their tissues, they do not develop Huntington’s-like disease, and breed normally, producing approximately 50% hemizygous offspring in each litter for 7-8 months or more. Ovarian transplantation, then, allows for much more efficient breeding performance from these colonies, resulting in significant savings in both mouse room shelf space and cost.
Ovarian transplantation overcomes genotyping difficulties
Historically, ovarian transplantation also has been used in managing strains containing spontaneous mutations that are either uncharacterized or for which molecular genotyping assays are not available. Homozygous B6C3Fea/a-Csf1op/J (000231) females (op/op), for example, fail to lactate, and homozygotes of both genders are extremely fragile; therefore, maintaining this strain by breeding homozygous mutants is not reliable. Homozygous mutant offspring can be identified easily from heterozygous (op/+) and wild type littermates by their phenotype (small size, domed skull, absence of incisors), but theop/+ littermates that are necessary for breeding cannot be visibly distinguished from their wild-type (+/+) siblings. The op mutation was identified in the 1970s, long before PCR-based genotyping assays were available, and the only way to genotype the op/+ and +/+ littermates was by a laborious progeny testing process. Fortunately, as with the Tg(HDexon1)62Gpb hemizygote females, the oocytes produced in the homozygote (op/op) females’ ovaries are viable, and we can transplant ovaries from op/op females into histocompatible, ovariectomized hosts. These ovarian transplanted females are then bred to wild-type males to produce obligate op/+ heterozygotes for breeding, which do not require genotyping or the labor-intensive progeny testing that would be required, otherwise, to identify them.
Ovarian transplantation improves colony efficiency
Ovarian transplantation, too, can be used to improve the efficiency with which a colony produces homozygous mutant mice. Indeed, at The Jackson Laboratory, we use ovarian transplantation to maintain our large B6.V-Lepob/J mice (000632) colonies because, although homozygous (ob/ob) females produce viable oocytes, they fail to regularly enter the estrous cycle, do not ovulate, and, therefore, do not breed successfully. Likewise, homozygous males are sub-fertile. By breeding ovarian transplantedob/ob females with unaffected, heterozygous (ob/+) males we can produce twice as many homozygous mutant offspring (50%ob/ob) as from ob/+ X ob/+ matings (25% ob/ob), dramatically improving our colony’s productivity. Further, becauseob/ob homozygotes are easily identified by their obese phenotype as early as 4 weeks of age, the ob/+ males that are necessary for breeding and that are used as controls for some experiments can be identified without genotyping. (We do still verify, however, the genotypes of the ob/+ males that we use for breeding to avoid breeding errors.)
Choosing the right ovary recipient
When maintaining a strain by ovarian transplantation it is important to make sure that the recipient’s ovaries are completely removed. If they aren’t, the recipient female may bear offspring derived from her own (wild type) ovaries instead of those derived from the homozygote female donor. To distinguish between these two scenarios, we use recipient females that carry a different, dominant, coat color allele that will not be carried in the offspring derived from the transplanted ovaries. Any offspring produced with the recipient’s coat color will have been derived from her residual ovaries, and can be easily identified and culled (Figure1).
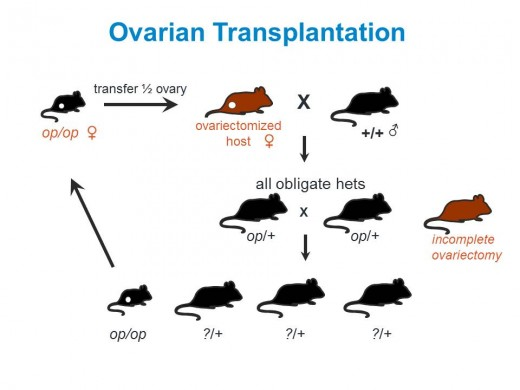
Figure1. Ovarian transplantation (OT) can overcome both breeding and genotyping difficulties. In this example homozygousop/op mice are infertile. Using OT, heterozygous op/+ mice for breeding can be identified without genotyping.
Alternative Strategies
A colony’s poor breeding performance might be overcome using strategies besides ovarian transplantation. For strains that produce litters regularly, but the females simply fail to take care of the pups once they are born, transferring the pups to the care of a foster mother may be the solution. For other strains,in vitro fertilization (IVF) might be the answer. Specifically, IVF can be used to create large cohorts of age-matched mice for experiments or for rapidly expanding a breeding colony’s size. Poor breeding performance is no doubt a mouse colonist’s greatest frustration. Fortunately, the techniques described here are available to help in overcoming them. It is true that both ovarian transplantation and IVF require technical expertise in order to perform them, but the techniques are widely used, and can be provided by the core facilities at many institutions. If you lack the personal or in-house expertise to perform these techniques, JAX offers courses where you can gain hands-on experience in learning the methods. Alternatively, our
JAX® Breeding Services group can save you the trouble and manage your troublesome colonies for you using whatever reproductive techniques are required to maximize their breeding productivity.Let us know if we can help!
