Happy or SAD: The chemistry behind depression
Blog Post | December 22, 2015

Depression is a serious condition that negatively affects how a person thinks, feels, and behaves. According to a recent study (Nature, 2014) more than 350 million people are affected by depression, making it one of the most frequent causes of disability and common disorders that affect humans worldwide.
What causes depression?
During the last decade, increased access to brain imaging technology has allowed neuroscientists and hospital clinicians to view the brain in detail, measure neural activity, and quantify neurotransmitter levels. Such studies have revealed many clues regarding the underlying contributing factors of depression and the pathophysiology of this disease.
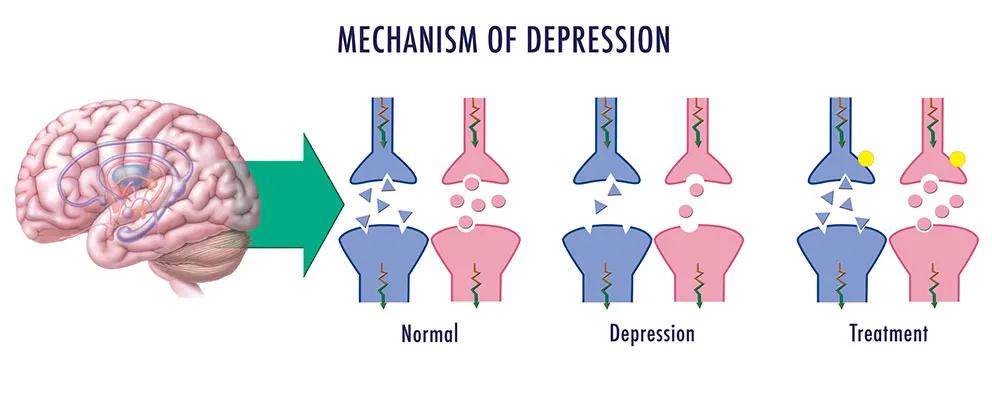
Serotonin: the happy neurotransmitter
Various animal models have demonstrated that chronic stress causes low serotonin levels in the brain. In patients, low brain serotonin activity correlates with a higher risk for more violent attempted and successful suicides. Serotonin levels have also been implicated in seasonal affective disorder (SAD).
Currently, the most widely prescribed antidepressants are selective serotonin reuptake inhibitors (SSRIs), which include brand names such as Prozac, Zoloft, Paxil and Celexa. These drugs block the uptake of serotonin (via SERT) by the neurons that produced it, making this neurochemical more available to neighboring postsynaptic neurons.
Dopamine: a chemical for motivation
Reduced dopamine levels, too, may contribute to depression. Although dopamine was thought initially to be crucial to the “reward” system in the brain and the neurons that control feelings related to pleasure, it more directly impacts the neurons that underlie motivation and habit formation. Dopamine motivates people to take action toward goals, desires, and needs, and issues a surge of reinforcing pleasure once they’ve been accomplished. Low dopamine levels make people and animal models less likely to work toward achieving a goal.
People with clinical depression often have increased levels of monoamine oxidase A (MAO-A), an enzyme that breaks down key neurotransmitters, resulting in very low levels of serotonin, dopamine and norepinephrine. In 2013, a team from Stanford using optogenetic mouse models showed thatinhibiting midbrain dopamine-releasing neurons induced depression-like behaviors caused by chronic stress, reinforcing the link between low dopamine and depression in patients.
Therapies for patients that don’t respond to standard treatments
Notions that the brain is a chemical soup in which not enough dopamine or too little serotonin contribute to severe depression helped pharmacologists design a series of antidepressants that work –although only in some cases. For too long, however, this view has limited advances in the field, including the delay to develop treatments for the 30% to 40% of depressed patients who, according to the National Institute of Mental Health, generally don’t respond to standard antidepressant treatments.
Ketamine: A promising quick fix
One of the most exciting, recent findings related to treatments for depression was the discovery that ketamine, a glutamate NMDA receptor antagonist and a FDA approved anesthetic, appears to counteract depression in a matter of hours. Although ketamine may be an alternative treatment for many patients with major depressive disorder who don’t get better with standard antidepressant drugs, it hasn't been thoroughly studied for long-term safety and effectiveness, and the FDA hasn't approved it to specifically treat mood disorders. Nevertheless, someclinics are providing “off-label” treatments. In the meantime, pharmaceutical companies are spending millions to develop patentable derivatives and ketamine analogues.
Depressive environmental factors
The biomedical community has come a long way towards understanding what causes depression and how best to treat some individuals- but they still don’t have answers for the most effective ways to treat all of the different disease subtypes. We cannot ignore the fact, too, that trauma early in life, hormonal changes, sustained stress, or sudden, emotional losses can push some people over the edge.
Interestingly, alterations in brain wave electrical patterns during sleep (for example, reduced deep sleep and time spent dreaming) and changes in the cerebral blood flow, also have been linked with depression.
Depression: A public health problem flying under the radar
Despite indications that depression is a predictor of aggressive behavior towards oneself or towards others, many people seem to ignore this, and prefer to sweep this problem under the rug. Why? One of the reasons may be that for many years, scientists couldn’t explain the physiological basis of the disease (like the brain lesions and plaque deposits that, for example, are commonly observed in patients with advanced Alzheimer’s disease). Although depression is thought to affect nearly 10% of the US population, and as many as two thirds of those who commit suicide exhibit some symptoms of depression, society has thus far been slow to react to this public health concern.
New hope to treat mood disorders
Given the complex and variable nature of mood disorders, outcomes likely will be better with treatments that are individualized and multi-faceted. After a period of belt tightening, last week Congress overwhelmingly approved a 2016 spending package that includes a $2 billion increase for the NIH.
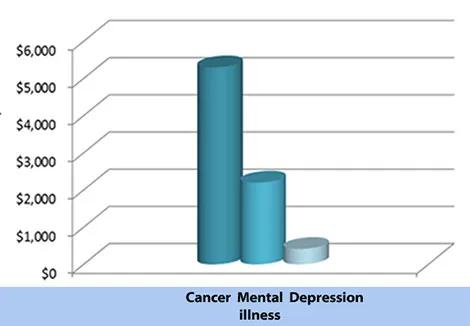
There is hope that in the near future some of the grant funding practices that have advanced our better understand or and treatments for cancer can be applied toward an effort to better understand and treat depression. Within the last decade, NIH funds have supported incredible strides forward in oncology, including the widespread adoption of tumor marker biology in assessing an individual’s risk for developing cancer and advances in personalized medicine for treating patients (Figure 2). Investing more resources in laboratories and hospitals that study and treat mental illness will transform our understanding of depression and lead the way toward new strategies that support the prevention and recovery of this disease.