The hardest thing to see is what is in front of your eyes.
– Johann Wolfgang von Goethe
That Goethe quote was taped to one of my mentor’s microscopes and I thought of it when a recent article characterizing a mouse model for liver disease caught my eye. The pathology was drop-dead gorgeous, the characterization went incredibly deep, and the introduction suggested that the model was found serendipitously. I had to know more.
Serendipity is more than luck…way more.
It takes wisdom to recognize unsought value; this is precisely what makes serendipity far more singular than mere luck. Luck requires no experience or skill. Serendipity does, and thus holds in science a particular allure. Many discoveries arise through serendipity. Just over a decade ago, the Institute for Laboratory Animal Research (ILAR) celebrated this fact by dedicating an entire issue of their journal, the ILAR Journal, to the concept of Serendipity, Science, and Animals.
The articles published therein reflect and speculate on the role of serendipity, defined broadly for the purposes of the issue as “the rational exploitation of chance observation, especially in the discovery of something useful or beneficial (Campbell, 2005).” The issue covers many species and stories, including an article written by JAX’s Professor Emeritus, Muriel T. Davisson on how serendipity was critical to the discovery of many important spontaneous mutations in mouse colonies maintained at The Jackson Laboratory (Davisson, 2005). The NOD (non-obese diabetic) inbred mouse, also, is a great serendipity story (Mathews and Leiter, 2005).
Serendipity may be misunderstood as a research strategy, perhaps due to negative connotations associated with the word’s usage as a synonym for luck (Stoskopf, 2005). Nevertheless, part of finding great animal models is recognizing them when they come along. This article highlights the characterization of a clinically comprehensive mouse model of non-alcoholic steatohepatitis (NASH) and underscores the importance of experience in serendipity-based scientific discovery. Dr. Sanyal and his collaborators deserve considerable credit for not only recognizing this model, but undertaking the stewardship needed to validate it thoroughly.
Quality takes time
One of the most frustrating truths that I faced as a student of comparative pathology was the gap between the conceptual descriptions of disease pathogenesis that I had learned and the actual appearance of diseased tissue in front of me. I would fixate and agonize over the identity of a single cell in the corner of a slide, missing the juicy pyogranulomas exploding in the center of the field of view, like a clueless kid oohing and aahing at a weird rock on the ground while standing at the lip of the Grand Canyon.
Even after my teachers had sufficiently coached me on basic anatomy and common lesions, I developed the bad habit of jumping to conclusions. “Take your time” they implored, “Look at the whole slide first! Then prioritize the findings. Then describe them (“Describe! Not interpret!”). Describe. Then, and only then, interpret.”
Yes, “take my time” but please hurry up and submit my already-late reports, I would grouse to myself, and go back to squinting into my eyepieces and thinking about the fact that all the answers were right in front of me. That knowledge, however, was little comfort when I was just too inexperienced to see them!
Later, as a mentor myself, I would ache with empathy for my students embarking on their own educational journeys to learn comparative pathology and histology. “Patience young type-A grasshoppers: mastery of a subject takes time!” When years of work go in to a creative effort—it shows.
The beginning
“As I tell everyone, I was involved in NASH when was there no field of NASH…it was just a single blade of grass.” –Dr. Arun Sanyal
Dr. Sanyal first began to publish on links between NASH and insulin resistance in the early 2000s (Sanyal, 2001; Choudhury and Sanyal, 2005). Nonalcoholic steatohepatitis (NASH) is a common, chronic liver disease characterized by fat infiltration with or without inflammation that can progress to fibrosis or cirrhosis, or both. Currently, no therapies for treating the disease have been approved.
“So many times people cure diseases in mice…and then are never able to replicate that in humans,” Dr. Sanyal explained, kindly taking time from his busy schedule to talk with me about the serendipity story behind his model. An issue with many mouse models (not just models for NASH) is that diseased tissue phenotypes often do not share the same pathogenesis as the human disease.
Early on his group recognized limitations in the available NASH models that could delay progress through misleading results. They started by asking, “What is missing? Why don't we like the existing models?” They specifically identified the following limitations:
- Involvement of gene knockouts not associated with human NASH
- Absence of insulin resistance
- Induction by non-physiological dietary alterations (abnormal levels of cholesterol)
His group then searched for a model with defined characteristics that would make a better preclinical model of NASH. Their ideal model would share the same characteristics as the human disease:
- Cause - too many calories
- Risk factors – obesity, insulin resistance, dyslipidemia
- Histology, disease outcomes, gene expression
- Hepatocellular carcinoma that arises as part of the natural disease progression and does not require induction by a carcinogen
DIAMOND in the rough
According to Dr. Sanyal, a colleague, Dr. Sandra Erickson, Ph.D. at the University of California, San Francisco, first brought his attention to the mouse. “She was looking for some mice to develop and study gallstone disease,” he said, and had found one that tended to accumulate fat in the liver, and even a little bit of scar tissue.
Cirrhosis, - the end-stage state of a liver chronically inflamed - is an aspect of NASH that was not easily modeled, so Dr. Sanyal took the mice offered by Dr. Erickson and began to work with them. As their work progressed, Dr. Sanyal sent slides to some well-established pathologists in the U.S., but received apologies that they were too overwhelmed with their human caseload to take the time to look at them. Later at a meeting, Dr. Sanyal found himself in a conversation with Dr. Pierre Bedossa, M.D., Ph.D., a very highly respected pathologist at Hospital Beaujon in Paris, France, and convinced him to take a look. After shipping the slides, Dr. Sanyal said that Dr. Bedossa called him the very next day asking, “What mice are these? Where did you get them? What did you give them? I've never seen mice that look like this. These look…almost like human disease."
Dr. Sanyal explained that the liver phenotype of the mice was inconsistent initially, but stabilized with further inbreeding. When they were sure that tumors developed predictably, they found collaborators and sent samples for transcriptomic analyses. He said, “Once we found that at the transcriptomic level there was pretty good concordance, then we got really excited.”
Time to shine
The DIAMOND mice were created from inbreeding B6129SF2/J (Stock# 101045) mice - that is, by breeding them sister X brother exclusively for 20+ generations - to produce a mouse with a novel genetic background. The NASH phenotype can be induced in adult male mice (8–12 weeks of age) by offering ad libitum a “Western diet” characterized by high fat (42% kcal from fat), high carbohydrate, , and containing 0.1% cholesterol , paired with drinking water containing a high fructose-glucose solution. Control mice are fed a standard chow diet and water without sugar. Disease progression over a DIAMOND animals’ lifetime is shown in Figure 1, below.
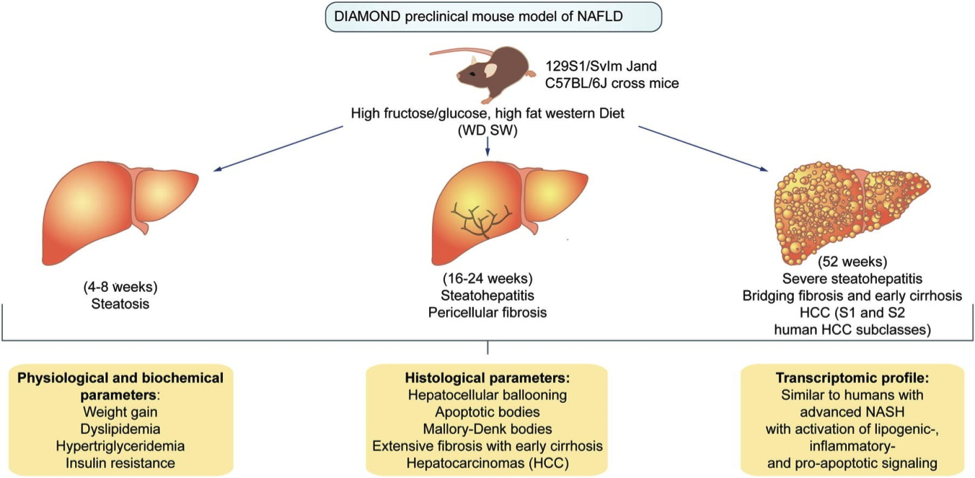 |
|
Figure 1. NASH disease progression in DIAMOND mice (Adapted from Asgharpour et al., 2016). |
Nothing sparkles more than beautiful pathology
When asked about the high quality of the histology (see Figure 2, below), Dr. Sanyal replied, “For us I think it was very important when we wrote the paper that the quality of the histology had to be such that anybody could look at it and say, ‘Oh, I see what you're talking about’.” He went on, “When you look at many of the papers in the literature, you know, people say, ‘We gave a NAS [nonalcoholic fatty liver disease activity] score’ and you look at the histology picture, and say ‘I can't see anything... other than fat... I can't tell anything.’”
Yes, “I can’t see anything in here!” That brings back memories: the plaintive cry of both a new student of histology as well as an expert looking at crappy histology. Gaps between conceptual descriptions and the actual appearance of disease tissue in the microscope don’t only afflict beginners.
He continued, “So we wanted to be sure that even the biggest naysayer could pick up that paper, look at it, read it, and say, ‘Oh, I see - this is what you mean by a balloon cell. This is what you mean by Mallory Body’. We wanted to show that what you are seeing in the mouse in terms of histology…it looks pretty much like what you see in humans.”
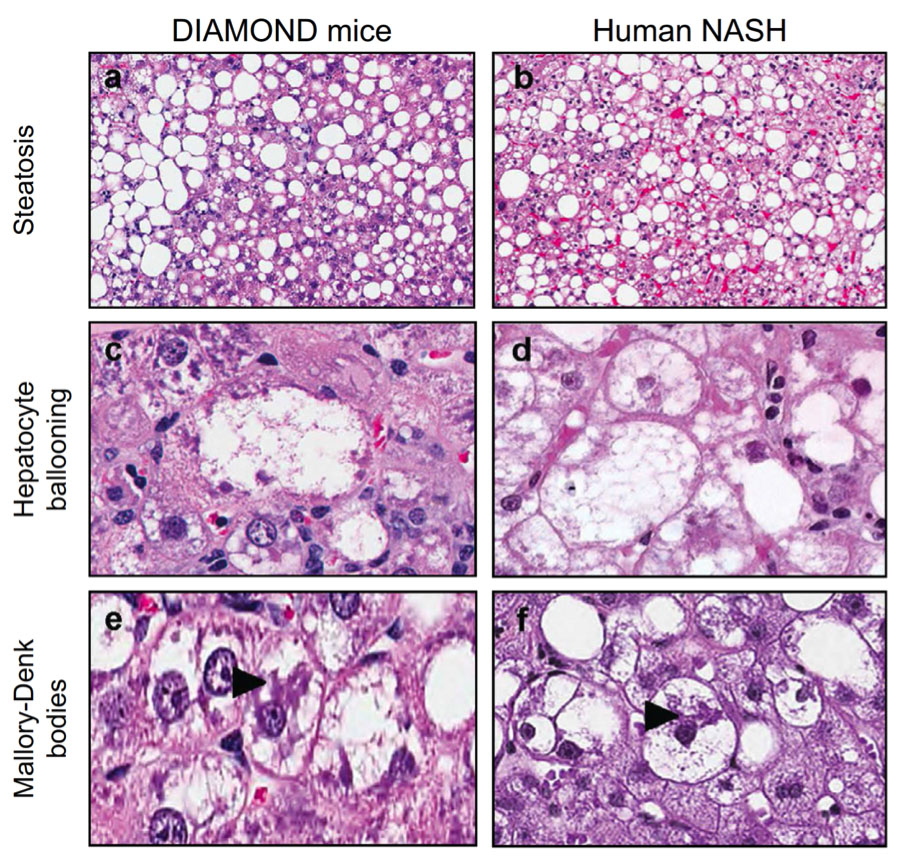 |
|
Figure 2. Histopathological comparisons between samples from a DIAMOND mouse model and a patient with NASH. Representative images of liver histology from DIAMOND mice (a,c,e) or human NASH (b, d, f) depicting steatosis (a, b; H&E; original magnification, 5X), Hepatocyte ballooning (c, d; H&E; original magnification, 40X), Mallory-Denk bodies (large arrow) (e, f; H&E; original magnification, 40X) (Adapted from Asgharpour et al., 2016). |
DIAMOND Mice: Past and Future
When asked to reflect upon the study, Dr. Sanyal replied, “It was a lot of work, but… it is better to take your time and get it right, so that whatever you put out there is something that everyone can ‘take to the bank’. Having a mouse where you control when they develop steatosis ...then steatohepatitis... then progressive fibrosis and then cancer....it allows us to model the disease at a molecular level and really do some in-depth analysis to understand ‘What are the molecular pathways driving the disease forward?’ That may allow us to actually develop a transcriptomic model of the disease, of disease development and progression analogous to what is known for many types of cancers. This may provide a basis for development of personalized medicine approaches in the future.”
Dr. Sanyal has also started a company, Sanyal Biotechnology and plans to take what has been learned from developing the DIAMOND mouse and improve models for alcoholic liver disease, another major unmet need.
Summary
A decade’s worth of effort is condensed into a clearly articulated clinic-pathologic presentation of the DIAMOND mouse model for NASH. DIAMOND mice are distinct from other NASH models with respect to the high number of clinicopathologic measures concordant with the human disease. Results suggest that this model could be helpful for developing therapies, understanding the genetics of NASH, and better understanding disease pathogenesis.
Moral of the story
Pay attention, be curious, and deeply cultivate expertise in your field in order to be better prepared to recognize the possible value of unexpected chance observations.
References:
Asgharpour A, Cazanave SC, Pacana T, Seneshaw M, Vincent R, Banini BA, Kumar DP, Daita K, Min HK, Mirshahi F, Bedossa P, Sun X, Hoshida Y, Koduru SV, Contaifer D Jr, Warncke UO, Wijesinghe DS, Sanyal AJ. 2016. A diet-induced animal model of non-alcoholic fatty liver disease and hepatocellular cancer. J Hepatol. 2016 May 31. pii: S0168-8278(16)30190-8. doi: 10.1016/j.jhep.2016.05.005. PMID: 27261415
Campbell WC. 2005. Introduction: serendipity in research involving laboratory animals. ILAR J. 46(4):329-31. PMID: 16179739
Choudhury J, Sanyal AJ. 2005. Insulin resistance in NASH. Front Biosci. 2005 May 1;10:1520-33. PMID: 15769642
Davisson MT. 2005. Discovery genetics: serendipity in basic research. ILAR J. 2005;46(4):338-45. PMID: 16179741
Mathews, Clayton E. and Leiter, Edward H. 2005. In Joslin's Diabetes Mellitus: Edited by C. Ronald Kahn. 14th Edition. Chapter 18 Rodent Models for the Study of Diabetes. pp292 – 302.
Sanyal AJ. 2001. Insulin resistance and nonalcoholic steatohepatitis: fat or fiction? Am J Gastroenterol. Feb;96(2):274-6. PMID: 11232664
Stoskopf MK. Observation and cogitation: how serendipity provides the building blocks of scientific discovery. ILAR J. 2005;46(4):332-7. PMID: 16179740
Rough diamond photograph courtesy of the U.S. Geological Survey. Original source: USGS "Minerals in Your World" website. Direct image link: [1] https://www2.usgs.gov/visual-id/credit_usgs.html#copyright