
Recently at his State of the Union Address, President Obama pledged a $215 million investment in the 2016 Budget to launch a new Precision Medicine Initiative. The Initiative is focused on advancing the research and development of patient-tailored strategies aimed at revolutionizing how we combat common diseases such as diabetes, neurological disorders, and cancer. The goal is to incorporate techniques and sophisticated models commonly used in science and apply them to directly improve patient care based on individual genomic and proteomic scenarios. With the human genome sequenced, high-throughput molecular screening technologies in place, and bioinformatics tools for metadata analysis developed, the opportunity to apply precision medicine universally to patients is now more possible than ever before. As new funding will soon stimulate growth of this field, next-generation small animal models will undeniably be instrumental in the development and testing of data-driven personalized therapeutics to fight some of the world’s most common diseases. (See video below)
Which diseases will be targeted in the Precision Medicine Initiative?
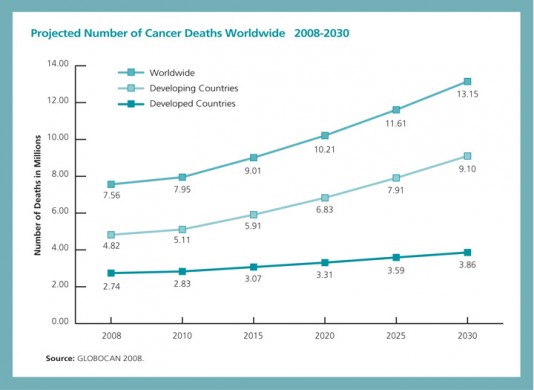
Although the initiative will focus on developing personalized treatment options for a number of common diseases, cancer is the number one player in this arena. Cancer is responsible for 1 in every 8 deaths worldwide, and as the most genetically diverse disease, cancer is likely to benefit the most from a personalized therapy approach. As Dr. Robert Weinberg discusses in this essay, each cancer diagnosis presents a unique disease due to different genetic alterations present between individual patients, between individual tumors, and even within a single tumor.
Additionally, cancer is a moving target, as it constantly changes molecularly. Too often patients in the clinic develop resistance to once-efficacious therapies, leaving them and their oncologists searching for alternative treatments. For these reasons, there is great promise for strategies that take into account individual variations, and are the most plausible for a disease that affects more and more people each year.
Projected Number of Cancer Deaths Worldwide
For each of these cancer patients, a different genetic signature is present, highlighting the need for a more personalized approach to combating this disease.
Avatars- miniature platforms for testing personalized therapies to treat cancer
The goal of precision medicine is to improve patient care through the integration of multiple approaches that scientists use routinely, such as the use of animal models, bioinformatics, and high-throughput sequencing. In particular, mouse models have been instrumental in the development of so-called avatars, which are platforms used to measure the efficacy of experimental treatments using a patient’s own malignant cells (either tumor fragments or other sources of primary tumor cells).
While these models have been around for a few years, costs and the time required to generate these models have made them largely inaccessible to the greater public. In general, avatars can cost up to hundreds of thousands of dollars, and the time to create a model can range from 9-12 months, which is not feasible for a patient who has exhausted standard of care options. However, novel approaches to address cost and time investment limitations will certainly make them more readily accessible to patients undergoing treatment.
Predicting patient response using Patient-Derived Xenografts (PDX) models
One such model used as a platform for validating cancer therapies is the NSG, or NOD.Cg-PrkdcscidIl2rgtm1Wjl/SzJ (005557) mouse engrafted with primary patient tumor fragments, known as PDX mice from the JAX PDX Resource. As the most versatile immunodeficient strain capable of engrafting the widest range of human cells and tissues, NSG mice have bolstered the field of predictive modeling to allow for more accurate representation of human tumors and responses to treatment.
JAX PDX mice are already demonstrating the potential for use as avatars for predicting patient responses to treatment. An example of this comes from the establishment of an EGFR L858R positive lung adenocarcinoma that initially responded to Tarceva (erlotinib) treatment in the patient, but later developed resistance (see figure below).
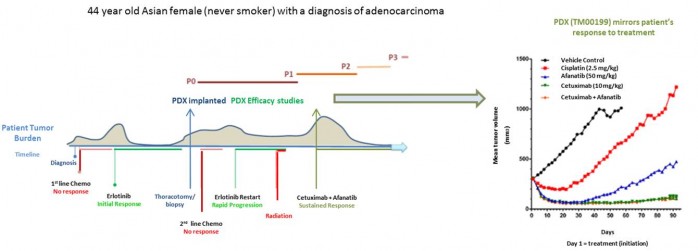
Patient tumor fragments were engrafted into NSG mice while the patient underwent combination Erbitux (cetuximab) and Afatinib therapy, which was successful in reducing tumor growth. Next-generation sequencing of the PDX tumor predicted erlotinib resistance, and in vivo trials with erlotinib validated resistance in the PDX model. Subsequently, cetuximab and Afatinib combination therapy was administered to the PDX model, which mirrored tumor growth inhibition observed initially in the patient.
By combining high-throughput sequencing technologies with avatar modeling, these experiments validate the concept of integrating genetic analysis and small animal models as avatars to predict patient treatment responses and to test individualized therapies without putting patients at risk in the process.
JAX Genomic Medicine’s role in advancing Precision Medicine
In addition to PDX models serving as patient avatars for drug efficacy testing, progressive health care institutions are using genetic and molecular screening of patient-specific tissues to serve as a guide for more individualized and targeted treatments. JAX Genomic Medicine has teamed up with cancer centers such as UC Davis Comprehensive Cancer Center and Beth Israel Deaconess Medical Center to develop new treatment strategies and to use JAX PDX models side-by-side with patients undergoing treatment .
Working alongside clinicians, our team of molecular biologists, scientific curators and bioinformatics experts transform next-generation sequencing into actionable guidelines for oncologists to prescribe better approaches for patients using drugs that are already on the market, or those in clinical trials. Certainly in the near future, combining genetic analysis with PDX modeling will transform cancer treatments worldwide and most importantly, improve their quality of life and chances of survival .
Interested in testing your compound with our genetically defined low passage PDX models?
Check out the JAX PDX Resource to view comprehensive information about our patient-derived tumor bank of more than 350 models, including molecular characterization, immunohistochemistry, tumor growth rates, and standard-of-care treatment responses.
The promise of precision medicine
Watch Dr. Jo Handelsman, Associate Director for Science at the White House Office of Science and Technology Policy, explaining the importance of precision medicine. (Video produced by The White House).