Which comes first, synapse loss or dendritic remodeling during glaucoma?
December 21, 2015The possible role of synaptic pruning in glaucoma
Retinal ganglion cell (RGC) death is a hallmark of the progressive neurodegenerative disease glaucoma. In addition to dendritic arbor remodeling, soma shrinkage, and axonal atrophy, recent evidence suggests that presynaptic contacts on the ganglion cells may be lost prior to these events and contribute to the earliest stages of RGC dysfunction. To investigate this phenomenon further, a group of researchers led by Dr. Tatjana Jakobs of Harvard Medical School and Dr. Gareth Howell of The Jackson Laboratory recently published a series of experiments inPLoS One examining the distribution and frequency of excitatory presynapsic arbors on RGC cells in a mouse model of glaucoma and in wild-type mice following optic nerve crush.
Dendritic remodeling and synaptic pruning are evident in the DBA/2J mouse model of glaucoma
DBA/2J mice (000671) typically develop moderate to severe eye abnormalities that mimic human hereditary glaucoma by 11 months. In order to examine the distribution of presynaptic excitatory inputs on glaucoma-affected RGCs, the Harvard and JAX investigators injected DBA/2J and glaucoma-resistant controls (DBA/2J-Gpnmb+/SjJ (007048)) intravitreally with an adeno-associated viral vector (AAV) directing expression of a fluorescently labelled PSD-95, a scaffold protein that localizes to and labels postsynaptic densities. Two weeks after injection, retinas were harvested and stained with an anti-SMI32 neurofilament antibody, which preferentially labels α-ganglion cells. The latter treatment was necessary to discern PSD-95-GFP puncta that were localized to the RGCs. In the analysis, RGCs from the DBA/2J mice showed a 30% reduction in dendritic arbor complexity compared to DBA/2J-Gpnmb+ controls. Changes in the presynaptic input linear density (i.e. the number of PSD-95-GFP puncta per cell/length of total dendritic arbor) showed a similar 28% decrease in DBA/2J RGCs when compared to controls (Figure 1).
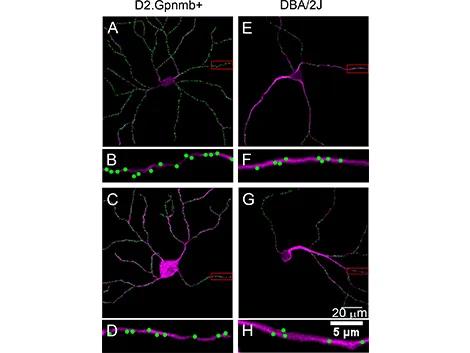
Dendritic remodeling and synaptic pruning are evident in the optic nerve crush mouse model
In order to examine RGC damage in a more time-controlled setting than is permitted by the spontaneous and progressive damage that occurs during glaucoma in DBA/2J mice, the Harvard/JAX team performed optic nerve crush on the left eye of C57BL/6J (000664) mice and examined the subsequent distribution of excitatory inputs on RGCs. Using the right eye as an internal control, the investigators observed decreases in dendritic arbors and a reduced synaptic density in optic nerve crushed-eyes compared to controls (48% by linear density quantitation). These findings mirror the results from the DBA/2J glaucoma experiments but with more drastic reductions in dendritic complexity and presynaptic inputs.
Synaptic pruning and subsequent dendritic remodeling occur almost simultaneously
Synaptic pruning is important in establishing and strengthening neural circuits during development, but it also plays a pathological role in neurodegenerative conditions. In glaucoma, synaptic pruning may be driven by components of the complement cascade that direct microglia to eliminate synaptic material, or through a "cell-autonomous" process in which changes within the RGCs themselves lead to synaptic loss and subsequent dendritic remodeling. Through either process, however, there should be a period when RGC dendrites are completely void of synapses, indicative of a pre-remodeling state of the dendrite.
Interestingly, the current study did not observe dendrites that were void of synapses in either the DBA/2J hereditary glaucoma mouse model or in the optic nerve crush model. This key finding makes it unclear whether presynaptic loss induces dendritic remodeling in glaucoma-affected RGC or is one of its consequences. Regardless, the MIT/JAX study indicates that whichever comes first – synaptic pruning or dendritic remodeling – the two phenomena proceed nearly simultaneously.
