Inhibition of obesity by manipulation of gut microbiota
It is increasingly evident that gut microbiota play a significant role in the development of metabolic disorders including obesity, diabetes and cardiovascular disease. Previous reports have shown that transfer of gut bacteria from lean mice into obese mice leads to reduced obesity phenotypes in the recipient mice. Therefore, gut bacteria may be an effective way to deliver therapeutic factors to reduce metabolic disorders. A recent article in Journal of Clinical Investigation (Chen, et al. 2014) illustrates the use of genetically engineered gut bacteria to delivery therapeutically relevant molecules that dramatically reduced body weight, adiposity and liver steatosis in obese mice
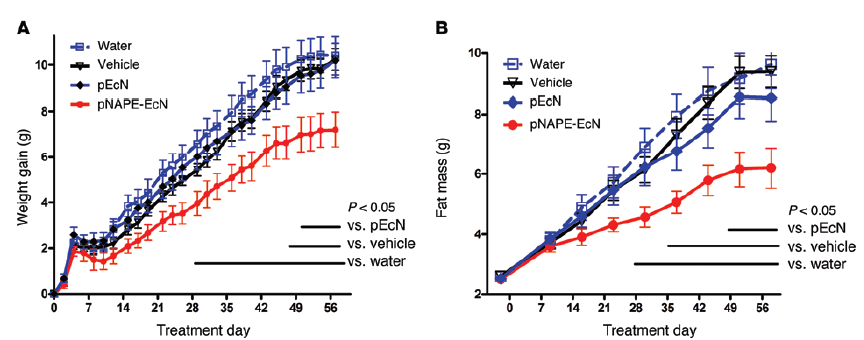
Treatment with bacteria containing pNAPE-EcN, but not pEcN, inhibits gain in body weight and adiposity. All values are the mean ± SEM (n = 10 mice per group). Solid bars indicate time points with significant differences between pNAPE-EcN and other groups (P < 0.05 by Bonferroni’s multiple comparison test). (A) Effect of treatments on gain in body weight from start of treatment (2-way RM ANOVA, for treatment P = 0.0073, for time P < 0.0001). (B) Effect of treatments on fat mass (2-way RM ANOVA, for treatment P = 0.0127, for time P < 0.0001).
Specially engineered NAPE-expressing E. coli. reduce obesity in mice
PN-acyl-phosphatidylethanolamines (NAPEs) are a type of natural lipid synthesized in the small intestine in response to feeding. NAPEs are then quickly converted into N-acylethanolamides (NAEs), which display potent anorexigenic properties. Direct administration of NAEs to diet-induced obese mice reduces food intake and obesity. Therefore researchers engineered E. coli, Nissle 1917 (EcN, wild type strain) to produce N-acyltransferase derived from Arabidopsis thaliana (pNAPE-EcN) at levels greater than normal and also greater than control bacteria (EcN transformed with empty vector, pEcN). In addition, insertion of a luciferase operon into both bacterial strains (pNAPE-EcN and pEcN) enables bioluminescent monitoring of gut colonization.
Supplementation of pNAPE-EcN bacteria to C57BL/6J (000664) mice in drinking water led to bioluminescence and therefore colonization in the intestinal tract. To test the effect of the bacteria on the development of obesity, glucose intolerance, and liver steatosis, C57BL/6J mice were fed a high-fat diet and given either the pNAPE-ECN or pEcN bacteria. pNAPE-EcN treated C57BL/6J mice maintained on a high-fat diet for 8 weeks gained significantly less body weight and adiposity as compared to pEcN and vehicle treated controls (Figure 1).
Additional metabolic symptoms alleviated by NAPE-expressing E. coli
In addition, compared to pEcN and vehicle treated controls, the pNAPE-EcN treated C57BL/6J mice displayed significantly:
- Improved glucose tolerance
- Lower plasma insulin and leptin levels
- Decreased cumulative food intake
- Decreased liver triglycerides and steatosis
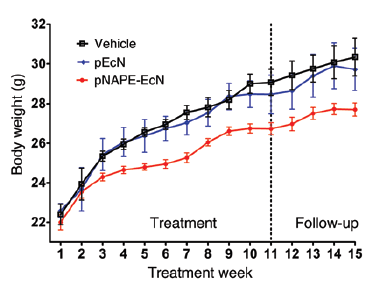
These improved metabolic parameters are not solely due to the decrease in food consumption by the pNAPE-EcN treated C57BL/6J mice. pEcN-treated mice gained significantly more weight and fat mass even when fed the same amount of food that the pNAPE-EcN-treated mice consumed. Eating a high-fat diet is not the only factor involved in human obesity; genetic factors also play a key role. Similar to humans, male TALLYHO/JngJ (005314) mice develop polygenic obesity on a standard diet and are therefore an important model useful for development of therapeutics. TALLYHO/JngJ mice treated with pNAPE-EcN also showed significantly less weight gain than pEcN and vehicle treated controls, as well as similar metabolic improvements as identified in the C57BL/6J mice (Chen, et al. 2014, Figure 13).
NAPE-expressing bacteria persist in the gut
pNAPE-EcN and pEcN treated mice excreted bioluminescent bacteria for 4 weeks after treatment ended, indicating persistence of the engineered bacteria in the gut. Body weight, fat mass and food intake in the pNAPE-treated C57BL/6J mice also remained significantly lower than the pEcN and vehicle treated controls even after treatment ceased. The difference in body weight continued to increase in magnitude until 6 weeks after treatment cessation. Similarly, body weight in the pNAPE-EcN-treated TALLYHO mice remained lower for at least 4 weeks after treatments ended.
These results support the hypothesis that genetically engineered bacteria can incorporate into the gut and provide sustained delivery of therapeutic molecules to inhibit chronic obesity and represents an effective alternative to daily dosing of therapeutic compounds.