If you research diabetes and/or obesity you may be wondering if the null mutation of the NAD nucleotide transhydrogenase (Nnt) gene (Toye et al. 2005; Freeman et al. 2006) in JAX® Mice strain C57BL/6J (B6/J, 000664) affects its responsiveness to diet-induced obesity (DIO). The answer is "No." JAX scientist Dr. Edward Leiter recently compared the Nnt allele's effects on DIO responsiveness in Nnt-mutant B6/J males to C57BL6/NJ males (B6/NJ, 005304), which have the wild-type Nnt allele (unpublished research, The Jackson Laboratory) (Leiter 2008).
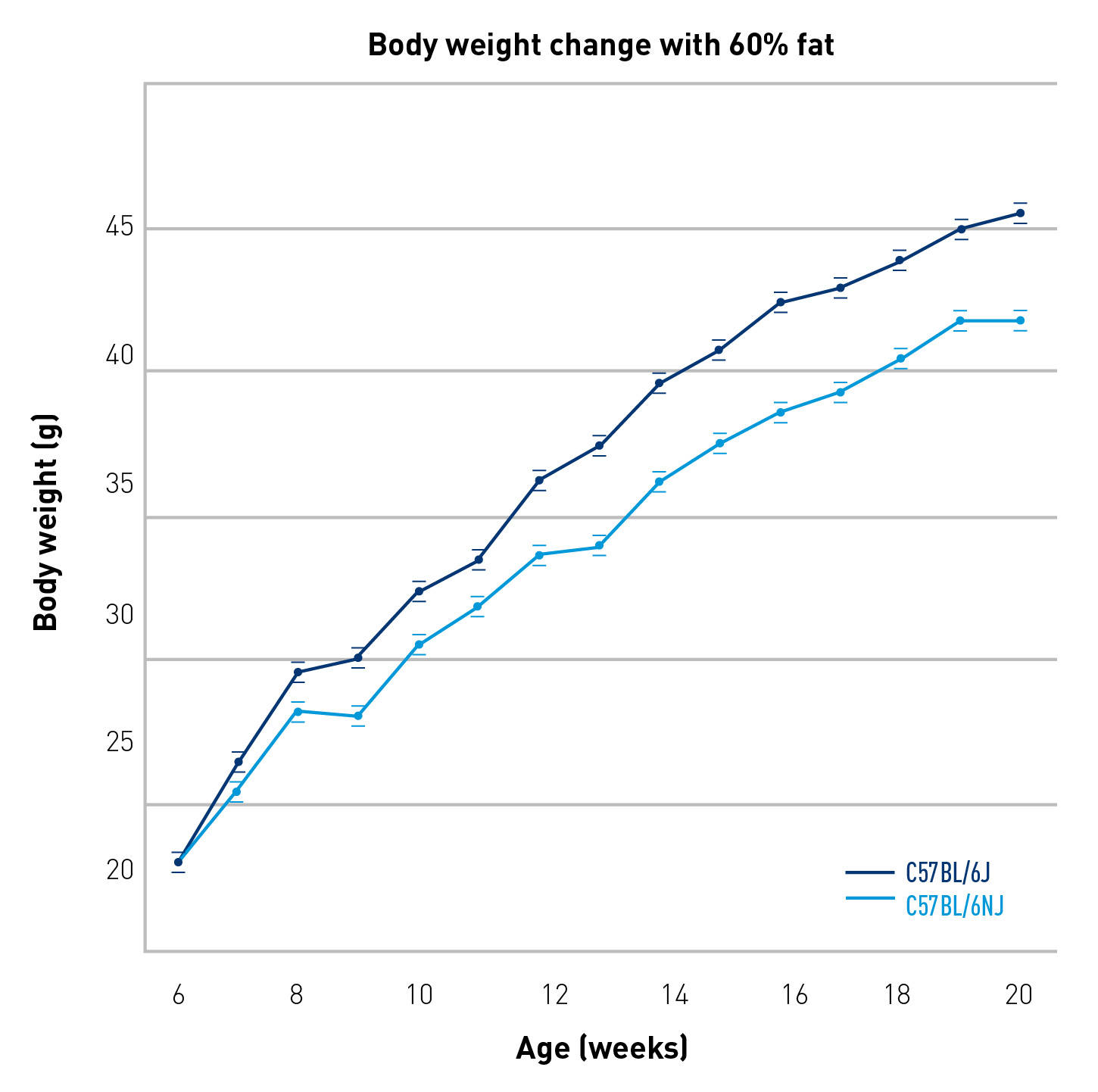
Cohorts of 40 six-week-old males from both substrains were fed a semi-defined diet containing 60% of its calories as fat (Research Diets D12492) for 14 weeks. The initial mean body weight (BW) of both cohorts did not differ, but, during the 14 weeks, the B6/J males significantly outgained the B6/NJ males, and, at the end of the study, weighed an average of 4g more than the B6/NJ males. Dual emission X-ray absorptiometry (DEXA) showed that this difference was due to a significantly higher lean and fat mass in B6/J males (Table 1). Mean non-fasting plasma glucose (PG) levels were also significantly higher in the B6/J males (sampled in eight, 12, 16, and 20-week-old mice).
Glucose tolerance became progressively more impaired in both substrains, but was significantly more severe in the B6/J substrain. Mean plasma insulin (PI) levels in 20-week-old mice were modestly (but not significantly) higher in B6/NJ males, consistent with their less severe IGT response (Table 1). Consistent with the greater adiposity of B6/J mice, mean plasma leptin (PL) levels were modestly higher in 20-week-old B6/J males and significantly higher in these males over time than in B6/NJ males (Table 1).
In summary, Dr. Leiter’s study revealed that B6/J males had a significantly more robust DIO response (higher weight gain, more adiposity, and more severe IGT) than B6/NJ males. Homozygosity for the Nnt mutation did not diminish male B6/J responsiveness to a 60% kcal fat diet. We cannot conclude that the mutant B6/J Nnt allele is the factor responsible for the more robust DIO response because certain strains that are even more DIO-responsive than B6/J, such as NON/ShiLtJ (002423), are Nnt wild-type (JAX® NOTES 2006). Likewise, strains such as A/J (000646), which are DIO-resistant (Surwit et al. 1988), are also Nnt wild-type (Freeman et al. 2006 supplemental material). As in humans, glucose homeostasis in these inbred strains is controlled by multiple genes.
Table 1. Comparative DIO phenotypes of 20-week-old B6/J and B6/NJ males fed a semi-defined diet containing 60% of its calories as fat (Research Diets D12492) for 14 weeks, from six to 20 weeks old (Data show mean ± SEMa).
|
Substrain |
PG (mg/dl) |
BW (g) |
Lean Tissue (g) |
Fat Tissue (g) |
|---|---|---|---|---|
|
B6/J |
237 ± 5b |
45.4 ± 0.4 b |
22.5 ± 0.5c |
20.6 ± 1.1c |
|
B6/NJ |
198 ± 5 |
41.2 ± 0.4 |
20.8 ± 0.5 |
16.3 ± 1.1 |
|
Substrain |
Fat Tissue (g) |
Percent Fat |
PI (ng/ml) |
PL (ng/ml) |
|---|---|---|---|---|
|
B6/J |
20.6 ± 1.1c |
46.4 |
8.9 ± 1.3 |
90.1 ± 5.3 |
|
B6/NJ |
16.3 ± 1.1 |
44.0 |
10.7 ± 3.1 |
72.4 ± 8.5 |
a n = 39-40/group for PG and BW, 12/group for DEXA parameters, and 10/group for plasma analytes
b Substrain differences significant at P < 0.0001 (statistical comparisons by JMP ANOVA program)
c Substrain differences significant at P = 0.03 (statistical comparisons by JMP ANOVA program)
Figure 1. B6/J males had a significantly more rapid DIO response than B6/NJ males (n = 39-40/group, P < 0.0001 by
ANOVA).
References
Freeman HC, Hugill A, Dear NT, Ashcroft FM. Cox RD. 2006. Deletion of nicotinamide nucleotide transhydrogenase; a new quantitative trait locus accounting for glucose intolerance in C57BL/6J mice. Diabetes 55:2153-6.
JAX® NOTES. 2006. NON/LtJ Males: a new model of diet-induced diabesity. JAX® NOTES 503:4.
Leiter EH. 2008. Does a mutant NAD nucleotide transhydrogenase (Nnt) gene in C57BL/6J impair responsiveness to diet-induced obesity? The Jackson Laboratory.
Surwit RS, Kuhn CM, Cochrane C, McCubbin JA Feinglos MN. 1988. Diet-Induced type 2 diabetes in C57BL/6J mice. Diabetes 37:1163-7.
Toye AA, Lippiat JD, Proks P, Shimomura K, Bentley L, Hugill A, Mijat V, Goldsworthy M, Moir L, Haynes A, Quarterman J, Freeman HC Ashcroft FM, Cox RD. 2005. A genetic and physiological study of impaired glucose homeostasis control in C57BL/6J mice. Diabetologia 48:675-86