A mouse model for menopause with ovarian follicle depletion
JAX Notes | July 1, 2004
Researchers at The University of Arizona have developed a new mouse model of menopause. Using intraperitoneal injections of the industrial chemical, 4-vinylcyclohexene diepoxide (VCD), Patricia Hoyer, Ph.D., and Loretta Mayer, Ph.D., selectively depleted small primordial and primary follicles in the ovaries of mice. With the exception of the ovary, VCD administration has no generalized toxic effect on mice. Follicle pools in the ovary are nonrenewable; therefore, the result of VCD treatment is irreversible ovarian failure. In 2004, the authors demonstrated that by day 58, following a 15 day treatment protocol, all treated B6C3F1 mice are in persistent diestrus (acyclic). By day 127, significant differences between treated mice and age-matched cyclic controls include:
- reduced ovarian and uterine weights.
- elevated plasma luteinizing hormone and follicle stimulating hormone.
- reduced plasma progesterone and androstenedione.
- non-detectable levels of plasma 17β-estradiol.
It is believed that the interstitial cells of the human post-menopausal ovary contribute to androgen production. In the absence of 17beta-estradiol, the androgen/estrogen balance is altered. Mayer speculates that the excess androgen may contribute to pathologies associated with menopause. Mayer and colleagues have demonstrated that the VCD-induced follicle depleted mouse ovary produces androgen, mimicking the human condition. The VCD-induced mouse serves as a model for evaluating endocrine status in the residual ovarian (interstitial) tissue.
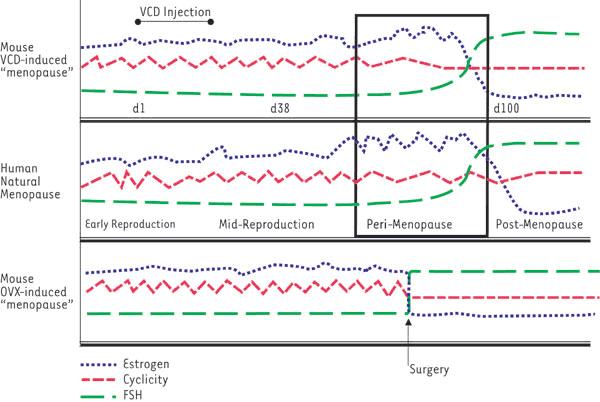
Figure 1. Comparison of changes in reproductive fluctuation between mice and women. Summarized patterns of cyclicity, estrogen levels and FSH levels measured in VCD-induced mouse "menopause" are compared with those values measured in human menopause and ovariectomized mice (OVX induced). Values for VCD-induced mice are assigned relative to the onset of dosing, day 1 (dl) with VCD for 15 days. Note the VCD-induced model mimics the human cycle.
Reference
Mayer LP, Devine PJ, Dyer CA, Hoyer PB. 2004. The Follicle-Depleted Mouse Ovary Produces Androgen. Biol Reprod Published online March 3, 2004, 1499890410.1095/biolreprod.103.016113.
