By Jens Rueter, MD and Andrey Antov, PhD, MBA
On behalf of all of the participating oncology practices in Maine, we are happy to share the latest progress updates for the Maine Cancer Genomics Initiative. To date, 150 patients have been registered and 59 clinicians at 10 sites are participating in the Study.
This month we feature Christopher Darus, MD. Dr Darus is the Principal Investigator for the MCGI Study at the Southern Maine Medical Center site and a member of the MCGI Clinical Steering Committee.

Christopher Darus, MD
Dr. Darus practices Gynecologic Oncology at Maine Medical Center. He holds board certification from the American Board of Obstetrics and Gynecology and specializes in Gynecologic Oncology. Dr. Darus received his MD from Wright State University School of Medicine, followed by a residency at University of Colorado Health Sciences Center and a Fellowship at University of Virginia Medical Center.
The 2018 MCGI Forum will be held April 6th - 8th at the Samoset Resort in Rockport. The conference runs from Fri April 6th (arrival 3pm and after) to Sun April 8th (networking and hotel check out by 10:30am). 6.5 hrs of AMA PRA Category 1 Credit™ and 6.5 ABIM MOC points will be offered for this event. All of the CME/MOC activities are scheduled on Sat April 7th. If you will be claiming ABIM MOC points at the Forum please remember to bring your ABIM member number (not state license number) as your points cannot be submitted without it.
This event will focus on the Promise of Precision Medicine and Experiences in Implementing Precision Medicine. Dr Rueter and Andrey Antov of the Maine Cancer Genomics Initiative will give an update on the status of the Initiative and the MCGI Study.
We will also have three nationally known speakers who will give presentations on New Genomic Signatures in Triple Negative Breast Cancer, Integration of Genomics in Clinical Pathways and Implementing the Precision Medicine Program.
Finally, there will be two breakout sessions in which clinicians and research staff at Maine oncology practices will review cancer somatic test ordering practices as well as somatic cancer testing implementation experiences.
Attendance is by invitation only. If you have questions about the Forum please reach out to us at [email protected].
The MCGI has scheduled more than 30 Genomic Tumor Boards sessions so far in 2018. Feel free to reach out to us at [email protected] for full list of GTB dates this year and details on CME offered for participants.
March:
April:
May:
Dates for Genomic Tumor Board sessions are subject to change. Contact us at [email protected] with any questions or for current list of session dates.
To learn more about MCGI, please visit our website at www.jax.org/mcgi, including biographies of Clinical Steering Committee members and our External Advisors. The Initiative is thankful to our External Advisors and Steering Committee members for all they do to support the Initiative. We are also thankful for the many Maine oncologists, clinicians and research staff participating in the Study.
By Pavalan Selvam, MBBS, Clinical Genomic Scientist
The JAX Action-Seq™ report contains a wealth of information about a patient's tumor testing results. Over the course of several newsletters, we highlight different aspects of the report and provide details that can help in interpreting the information provided. In this issue we present:
CLIA Educational Brief - Requisition and Pathology Report Review
Pavalan Selvam, MBBS, Clinical Genomic Scientist
Over the past few months, Analysis & Reporting CLIA briefs have touched on the quality control processes, variant classification system, as well as the molecular and clinical variant interpretation methods used to transform DNA sequencing data into the clinical test reports issued to physicians (Figure 1).
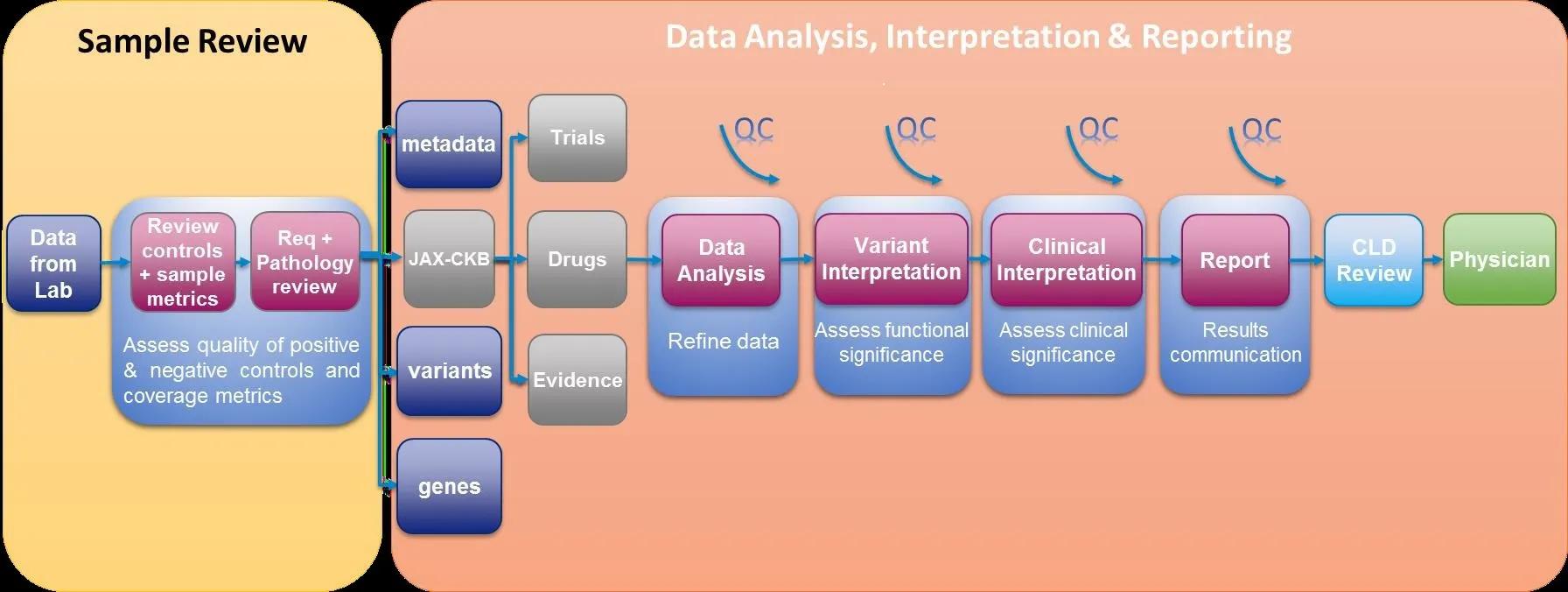
Another important component of this process is initial review of the medical history information gathered from the requisition, pathology report and other test result findings, which can fine-tune the relevance of clinical trials as well as the evidence for variants and therapies. Parameters such as hormone receptor status, previously identified genetic mutations, or other laboratory results that narrow focus of the somatic test results facilitates removal of non-eligible trials, inclusion/exclusion of known resistance mechanisms and higher specificity evidence to associate with detected variants during analysis and interpretation. We are happy to provide re-analysis of data upon request or further review variants with additional information that may influence variant classification.
As always; Information about test processing status is available by contacting Clinical Lab Customer Service, j[email protected]. The customer service direct line is 860-837-2162. If you have any additional questions related to technical or scientific aspects of the test, please do not hesitate to reach out to our Clinical Laboratory Director, Honey Reddi, PhD, FACMG at (860) 837-2062.
The 2018 MCGI Forum will be held on Friday, April 6th through Sun April 8th at the Samoset Resort in Rockport, ME. Registration for the Forum is open through Friday, March 30th; for the registration code please contact us at [email protected]. 6.5 AMA PRA category 1 credits and 6.5 ABIM MOC points will be offered for educational activities at the event.
Attendance is by invitation only. Lodging and meal costs will be covered for invited Maine clinicians and research personnel. Invitees may bring a spouse or guest; nominal meal charges will be assessed at registration for guests of invitees.
The group will have complimentary access to unlimited local and toll free calls, in room coffee, wireless internet in guest rooms and public space, access to the business center, outdoor zero entry pool, hot tub and fire pit, outdoor recreation including tennis courts, shuffleboard, basketball court and playground, concierge service, children's activities, and access to our state of the art full service health club including indoor pool, hot tub, steam saunas, strength room, group fitness room, cardio theater, and classes.
The Hartford Courant published an op-ed from JAX in the "Bright Ideas" special section by Sue Mockus about JAX's Clinical Knowledgebase (CKB)
An article in Hartford Magazine about "10 Great Biotech Advances" in Connecticut featured JAX's Clinical Knowledgebase, including an interview with Sue Mockus and a photo of Sue and her team. Please see page 60-61 for the story in the digital edition.
Edison T Liu, MD spoke at the San Antonio Breast Cancer Symposium in 2017 about the Tandem Duplicator Phenotype (TDP) in breast cancer. Dr Liu will be at the MCGI Forum this April to share a presentation on the TDP with the Maine oncology community.
The mission of the MCGI is to enable widespread access to clinical cancer genomic tests for the Maine oncology community and to increase the understanding of cancer genomics by Maine oncology clinicians. The Maine Cancer Genomics Initiative (MCGI), enabled through the generous financial support from The Harold Alfond® Foundation, leverages the strengths of key medical and bioscience research institutions in Maine to create and alliance focused on precision cancer diagnostics and treatment. The MCGI central office team is dedicated to ensuring your practice's engagement and experience with genomic cancer testing. Please feel free to reach out with any questions or comments. Email us at [email protected]