By Jens Rueter, M.D. and Andrey Antov, Ph.D., M.B.A.
On behalf of all of the participating oncology practices in Maine, we are happy to share the latest progress updates for the Maine Cancer Genomics Initiative. As of Dec 13th, 68 patients have been registered, 10 sites are participating in the Study and 7 Genomic Tumor Boards sessions have been run at 5 different locations. With the help of our collaborators from The Center for Outcomes Research & Evaluation at The Maine Medical Center Research Institute, Paul Han and his team, a poster was presented at the 2017 NNECOS Annual Meeting summarizing Initiative onboarding activities. The 2018 MCGI Forum will be held April 6th - 8th at the Samoset Resort in Rockport. You can find more details below in the events section. Attendance is by invitation only and registration information will be sent soon. If you have any questions about the Forum please reach out to us at [email protected]. In the past months we have had great success with MCGI Genomic Tumor Board sessions. These sessions will continue in 2018. You can attend in person or virtually as your schedule permits. A series of 24 sessions has already been scheduled and more will be added in coming weeks. Please see the sessions below for Jan and Feb. For a full schedule contact us at [email protected], calendar invites will be sent to the community in the New Year.
January
February
To learn more about MCGI, please visit our recently updated website, now including biographies of Clinical Steering Committee members and our external advisors. The Initiative is thankful to our External Advisors and Steering Committee members for all they do to support the Initiative. We are also thankful for the many Maine oncologists, clinicians, and research staff participating in the Study. In each newsletter we will feature a key MCGI community member in this Spotlight.
This month we feature Dr. Julia Moukharskaya. Dr. Moukharskaya leads MCGI study efforts as the Principal Investigator for the Harold Alfond Center for Cancer Care at MaineGeneral Hospital.

Dr. Moukharskaya is a medical oncologist employed at the Harold Alfond Center for Cancer Care in Augusta. A member of MaineGeneral Medical Center's active staff, she is certified by the American Board of Internal Medicine in Hematology and Medical Oncology. Dr Moukharskaya received her medical degree from the Belarusian State Medical University in Minsk, Belarus and PhD from the Republican Science-Practical Center also in Belarus. She completed a residency in Internal Medicine at East Tennessee State University followed by a fellowship in Hematology & Medical Oncology at the University of Vermont College of Medicine.
By Matthew Prego, Clinical Genomic Analyst
The JAX Action-Seq™ report contains a wealth of information about a patient's tumor testing results. Over the course of several newsletters, we will highlight different aspects of the report and provide details that can help in interpreting the information provided.
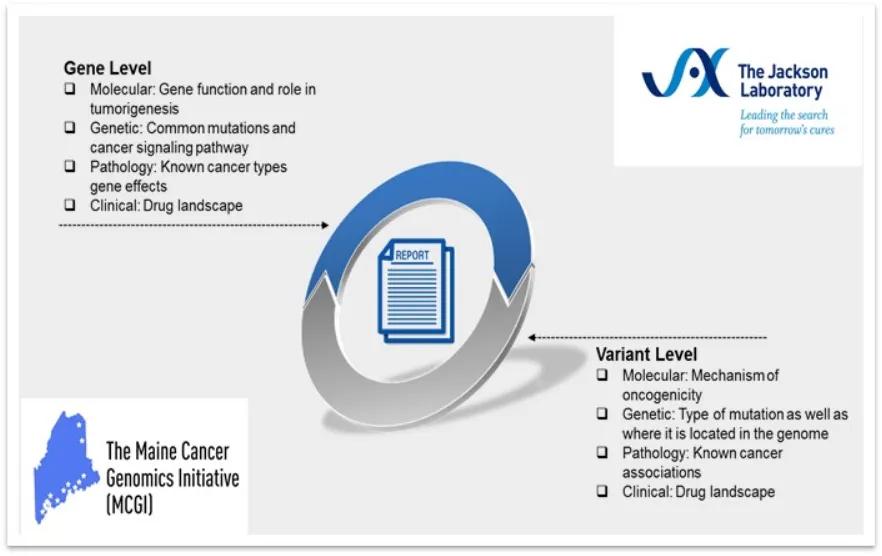
In this issue we present: CLIA Educational Brief - Overview of Gene and Variant Interpretation
All ActionSeq Plus reports contain gene and variant summaries based on interpretive findings. Gene descriptions cover fundamental clinical genetic information such as the encoded protein and its primary function, disease mechanism, common variant types, common tumors associated with aberrant forms of the gene and a brief overview of the therapeutic landscape. Similarly, variant descriptions include information such as the impacted protein domain, molecular function, pathogenic mechanism and tumors known to harbor the specific aberration (Figure 1). Evidence supporting the role of these genetic findings in cancer are collected from peer-reviewed literature, public databases and a variety of computational tools to assess clinical significance. Due to the rapid pace of oncology research, comprehensive re-assessment of variants is conducted on a 6-month cycle to ensure the most accurate and current information is provided in a clinical report.
Figure 1: Components of gene and variant information
As always; Information about test processing status is available by contacting Shannon Rowe in Clinical Lab Customer Service, [email protected]. Shannon's direct line is 860-837-2162. If you have any additional questions related to technical or scientific aspects of the test, please do not hesitate to reach out to our Clinical Laboratory Director, Honey Reddi, PhD, FACMG at (860) 837-2062.
By Kate Reed, MPH ScM, CGC
November Educational Offering
This month we are highlighting two resources from other organizations:
My Cancer Genome, developed by the Vanderbilt-Ingram Cancer Center, is a tool that matches cancer genetic variants to targeted therapies. This resource also provides information about clinical trials using genetically targeted therapies.
ASCO University's Molecular Oncology Tumor Board, a collaboration between the American Society of Clinical Oncology, the College of American Pathologists, and the Association for Molecular Pathology, are free (no ASCO membership required) monthly, user-driven discussions among cancer care providers. Topics are designed to help providers with interpretation and application of tumor molecular profiling tests.
Don't forget about these additional educational opportunities:
Free CME Modules available here.
Exploring Somatic Cancer Panel Testing is a self-directed module, which uses interactive cases to explore the benefits and limitations of large panel testing for different types of patients. You can earn 0.5 CME credits.
Interpreting Somatic Cancer Panel Testing Reports is a self-directed module, which uses interactive cases to explore and apply results from large somatic cancer panel tests. You can earn 0.5 CME credits.

The 2018 MCGI Forum will be held on Fri April 6th through Sun April 8th at the Samoset Resort in Rockport, ME. Attendance is by invitation only. Lodging and meal costs will be covered for invited Maine clinicians and research personnel. Invitees may bring a spouse or guest; nominal meal charges will be assessed at registration for guests of invitees. Registration for the Forum is open, please contact us at [email protected] for the registration code and visit here to register.
The group will have complimentary access to unlimited local and toll free calls, in room coffee, wireless internet in guest rooms and public space, access to the business center, outdoor zero entry pool, hot tub and fire pit, outdoor recreation including tennis courts, shuffleboard, basketball court and playground, concierge service, children's activities, and access to our state of the art full service health club including indoor pool, hot tub, steam saunas, strength room, group fitness room, cardio theater, and classes.
Dec 4th: Jens Rueter, MD and Paul Han, MD, MA, MPH presented an overview of the Maine Cancer Genomics Initiative at the annual meeting for The Hanley Center for Health Leadership in Freeport
Dec 11th: Jens Rueter, MD and Paul Han, MD, MA, MPH presented an overview of the Maine Cancer Genomics Initiative at the Steering Committee meeting of the IGNITE network.
_____________________________________________________________________________________________________________
The mission of the Maine Cancer Genomics Initiative (MCGI) is to enable widespread access to clinical cancer genomic tests for the Maine oncology community and to increase the understanding of cancer genomics by Maine oncology clinicians. The MCGI, enabled through the generous financial support from The Harold Alfond® Foundation, leverages the strengths of key medical and bioscience research institutions in Maine to create and alliance focused on precision cancer diagnostics and treatment.
The MCGI central office team is dedicated to ensuring your practice's engagement and experience with genomic cancer testing. Please feel free to reach out with any questions or comments. Email us at [email protected].