Humanized NSG mice are a gold-standard translational platform used for the evaluation of human immune cells in vivo, the evaluation of single and combinatorial drug approaches, and the production of predictive data for the clinic. These models can be used in immuno-oncology applications, stem cell biology, and preclinical efficacy testing. With hundreds of clinical studies, trials and scientific publications enabled by these models, you can confidently partner with The Jackson Laboratory for innovative, validated cell humanized mouse models and preclinical services expertise for your complex studies and research goals.
Immunomodulatory therapeutics commonly evaluated in humanized mice:

PBMC-engrafted humanized mice are an established platform for short-term testing of immunosuppressive drugs. Murine MHC I/II DKO models offer the additional advantage of enabling long-term studies without the negative effects of GvHD, while retaining robust T cell activation and functionality. When analyzing donor-specific toxicity (cytokine release) or tumor response, a key challenge is often the inherent variability in each patient's immune system—donors differ significantly in cytokine release levels, with some showing high release and others low. Tumor burden also varies by donor and does not always correlate with the severity of cytokine release syndrome (CRS), which can further complicate interpretation of results.
With JAX PBMC-engrafted humanized mice, you can choose from pre-characterized PBMC donor populations to suit your study, along with the added advantage of being able to reserve specific donor PBMCs to ensure more consistent and reproducible study results.
Donors C & E; high cytokine release & required high bispecific dose for TGI. Donor D; low cytokine release, but required high bispecific dose for TGI. Donors F & G; low cytokine release with TGI at lower doses (can tolerate higher drug dose).
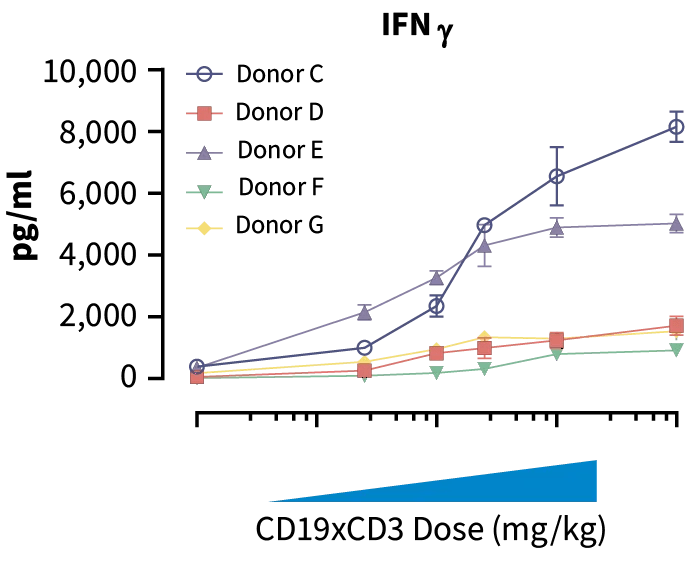
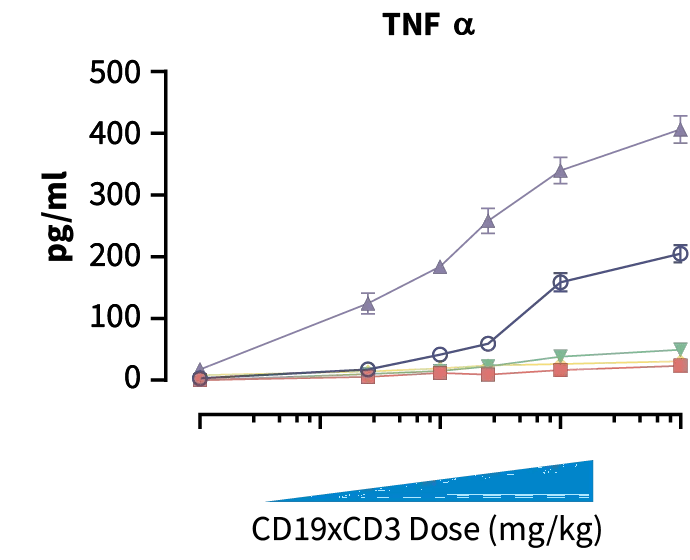
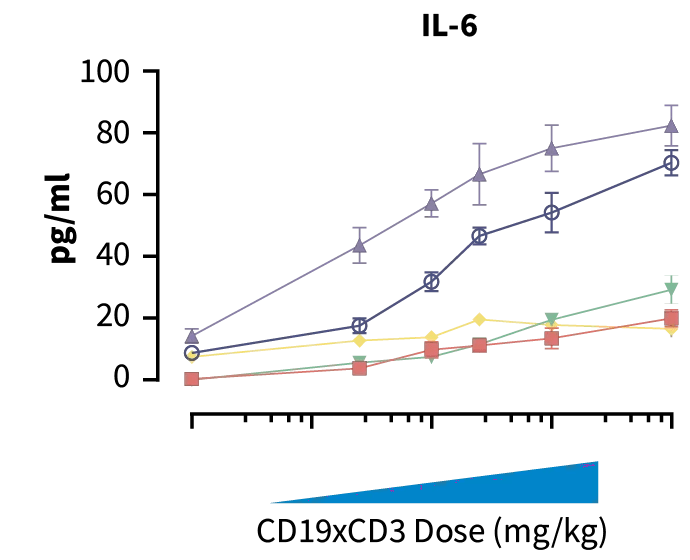
Increased dose of CD19xCD3 bispecific increased human cytokine concentration in blood. Donors C & E showed more elevated cytokine release than donors D, F, & G indicating high toxicity potential.
CD34+ engrafted NSG and NSG variants can be enrolled in a variety of different studies through our Preclinical Services team. These studies can evaluate the efficacy of various therapeutic strategies such as the depletion of host immune cells, CD19+ B cells, or the engagement of human immune cells to shrink engrafted tumor cells. In the study shown, CD34 huNSG-SGM3-IL15 mice were successfully used to evaluate a combinatory therapeutic response, as a measure of tumor burden and effects on tumor infiltrating lymphocyte (TIL) population, using an MDA-MB-453 tumor (low EGFR expression). In this model, the response to EGFR bispecific T cell engagers alone or to Avelumab (an anti-PDL-1 antibody) alone is significantly less than in mice treated with a combination of both. The population of immune cells in the TIL of harvested tumors is charactered by an increase in CD3+, CD4+ T cells, and a significant increase in CD8+ T cells. Due to the engineered IL15 cytokine in the Hu-SGM3-IL15 mouse, elevated CD56+ natural killer cells are seen in the TIL fraction and are present in various abundances based on treatment type.