Research probes a key contributor to glaucoma
Research Highlight
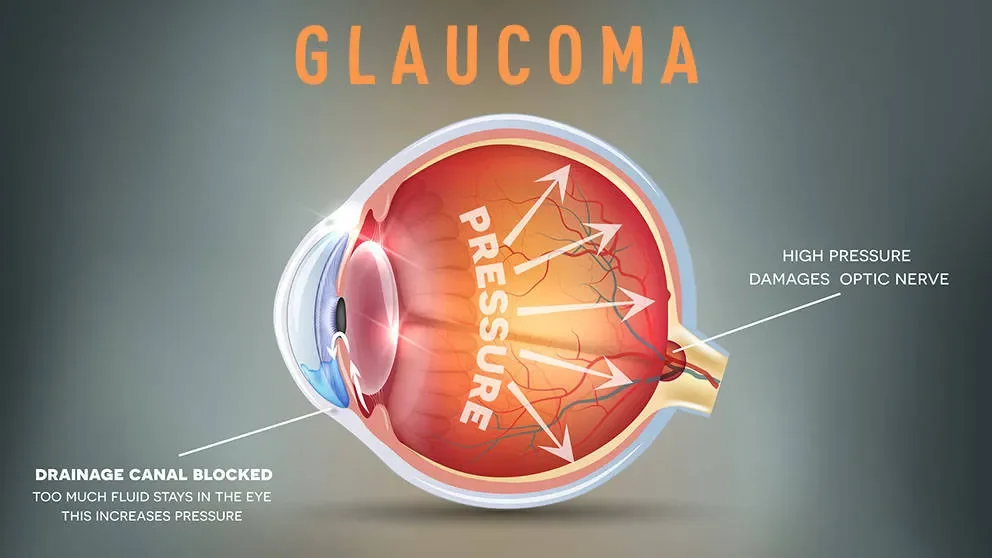
Glaucoma is the leading cause of irreversible blindness worldwide. On the surface, the disease appears relatively simple, with high pressure (intra-ocular pressure, or IOP) within the eye associated with the death of cells in the retina and optic nerve dysfunction. But the genetic and molecular mechanisms underlying the disease are quite complex. And even the exact role of pressure in disease progression has come under scrutiny, as sight can be preserved despite high IOP.
One of the factors now implicated in glaucoma-associated neurodegeneration is the endothelin (EDN) system, but the mechanisms are not yet known. The primary known function for EDN proteins is to constrict blood vessels, thereby raising blood pressure. They are part of a larger system of checks and balances that maintains proper vascular function, but if over expressed can contribute to hypertension and cardiovascular disease. In glaucoma, EDN signaling increases in retinal cells before any damage occurs, and blocking it lessens cell death. Furthermore, artificially overactivating EDN signaling in retinal cells kills them. So how does it all work?
JAX Associate Professor Gareth Howell, Ph.D., is teaming up with the University of Rochester Medical Center’s Richard Libby, Ph.D., to find out. Through a five-year National Eye Institute R01 grant totaling more than $2.6 million, they will investigate several aspects of how EDN induces neurodegeneration and cell death and how these mechanisms might be targeted therapeutically for early disease prevention, before nerve damage and cell death has occurred.
The work will focus on two distinct goals. First, Howell and Libby will determine exactly how the EDN system contributes to retinal cell death. For this, they will test genes that have already been implicated in cell death caused by glaucoma. Interestingly, they will also use genome-wide profiling of the cells’ transcription profile — the genes that have been transcribed to messenger RNA — to see what pathways are activated in the cells after EDN insult. This will lead to detailed new information about exactly what molecular signals are provoked and how they may contribute to cell death.
Second, they will determine which components of the EDN system are involved in both degeneration of the optic nerve and retinal cell death. The system has multiple receptors and signaling molecules, some of which have been shown to increase in activity in very early disease stages in a mouse model of glaucoma. Determining which components are critical for the damage — and how they do the damage — is essential for determining the specific mechanisms of the EDN system in detail. Howell and Libby hope that through this work they will determine the full potential for targeting the EDN system as a therapy for human glaucoma.

